CBSE class 9 Science Chapter 3 Atoms and Molecules notes in PDF are available for free download in myCBSEguide mobile app. The best app for CBSE students now provides Atoms and Molecules class 9 Notes latest chapter wise notes for quick preparation of CBSE exams and school based annual examinations. Class 9 Science notes on Chapter 3 Atoms and Molecules are also available for download in CBSE Guide website.
CBSE Guide Atoms and Molecules class 9 Notes
CBSE guide notes are the comprehensive notes which covers the latest syllabus of CBSE and NCERT. It includes all the topics given in NCERT class 9 Science text book. Users can download CBSE guide quick revision notes from myCBSEguide mobile app and my CBSE guide website.
9 Science notes Chapter 3 Atoms and Molecules
Download CBSE class 9th revision notes for Chapter 3 Atoms and Molecules in PDF format for free. Download revision notes for Atoms and Molecules class 9 Notes and score high in exams. These are the Atoms and Molecules class 9 Notes prepared by team of expert teachers. The revision notes help you revise the whole chapter in minutes. Revising notes in exam days is on of the best tips recommended by teachers during exam days.
Download Revision Notes as PDF
CBSE Class 9 Science
Revision Notes
CHAPTER – 3
Atoms and Molecules
1. Laws of Chemical Combination
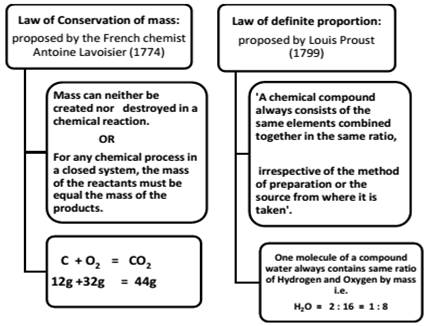
Limitation of “Law of definite proportion”
This law does not hold good when the compound is obtained by using different isotopes of the combining elements.
2. John Daltons Atomic Theory
Using his theory, Dalton rationalized the various laws of chemical combination which were in existence at that time. However, he assumed that the simplest compound of two elements must be binary.
3. Atoms, Molecules, Ions & Chemical Formula
Atom
An atom is the smallest particle of an element which can take part in a chemical reaction. It may or may not exist freely.
Each atom of an element shows all the properties of the element.
Molecule
The smallest particle of matter (element or compound) which can exist in a free state.
The properties of a substance are the properties of its molecules.
- MOLECULES OF ELEMENT : The molecules of an element are constituted by the same type of atoms.
- MOLECULES OF COMPOUND: Atoms of different elements join together in definite proportions to form molecules of compounds. (hetero atomic molecules)
- ATOMICITY: The number of atoms contained in a molecule of a substance (element or compound) is called its atomic
| Element | Formula | Atomicity |
| Ozone | O3 | 3 |
| Phosphorus | P4 | 4 |
| Sulphur | S8 | 8 |
| Oxygen | O2 | 2 |
- Based upon atomicity molecules can be classified as follows.
- Monoatomic molecules: Noble gases helium, neon and argon exist as He Ne and Ar respectively.
Diatomic molecules:
Triatomic molecules:
SYMBOLS
- The abbreviation used to represent an element is generally the first letter in capital of the English name of element.
Oxygen O Nitrogen N - When the names of two or more elements begin with the same initial letter, the initial letter followed by the letter appearing later in the name is used to symbolize the element
Barium Ba Bismuth Bi
Symbols of some elements are derived from their Latin names
| ELEMENT | LATIN NAME | SYMBOL |
| Sodium | Natrium | Na |
| Copper | Cuprum | Cu |
| Potassium | Kalium | K |
| Iron | Ferrum | Fe |
| Mercury | Hydragyrum | Hg |
| Tungsten | Wolfram | W |
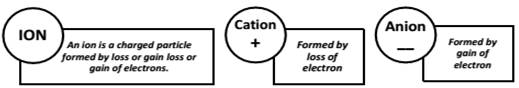
Polyatomic Ion : A group of atoms carrying a charge is as polyatomic ion.
Eg.
Valency : The number of electrons which an atom can lose , gain or share to form a bond.
OR
It is the combining capacity of an atom of the element.
- Chemical Formula : A chemical formula is a short method of representing chemical elements and compounds.
Writing a Chemical Formula -CRISS-CROSS rule
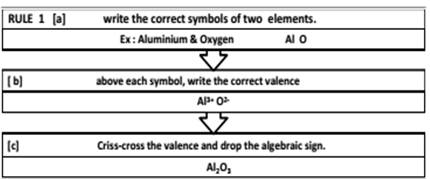



RULES > All subscripts must be reduced to lowest term (except for molecular or
Examples
| CATION | ANION | FORMULA | NAME |
| Aluminium sulphate | |||
| Ca | Calcium bicarbonate | ||
| Ammonium chloride | |||
| Sodium carbonate | |||
| Magnesium hydroxide | |||
| Sodium phosphate |
4. Mole Concept
The mole (mol) is the amount of a substance that contains as many elementary entities as there are atoms in exactly 12.00 grams of
The Avogadro constant is named after the early nineteenth century Italian scientist Amedeo Avogadro.
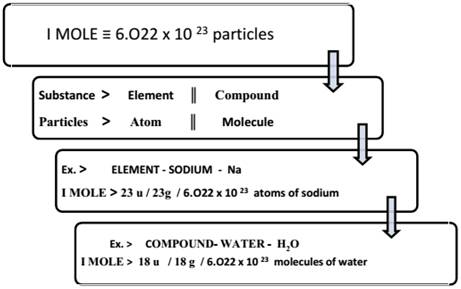
GRAM MOLECULAR MASS
Gram molecular mass is the mass in grams of one mole of a molecular substance.
Ex: The molecular mass of is 28, so the gram molecular mass of is 28 g.
ATOMIC MASS UNIT
An atomic mass unit or amu is one twelfth of the mass of an unbound atom of carbon-12. It is a unit of mass used to express atomic masses and molecular masses.
Also Known As: Unified Atomic Mass Unit (u).
MOLECULAR MASS : A number equal to the sum of the atomic masses of the atoms in a molecule. The molecular mass gives the mass of a molecule relative to that of the 12 C atom, which is taken to have a mass of 12.
Examples: The molecular mass of is approximately 30 or . Therefore the molecule is about 2.5 times as heavy as the 12C atom or about the same mass as the NO atom with a molecular mass of 30 or (14 +16).
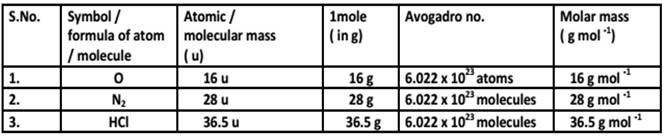
5. Molar Mass & Avogadro Constant
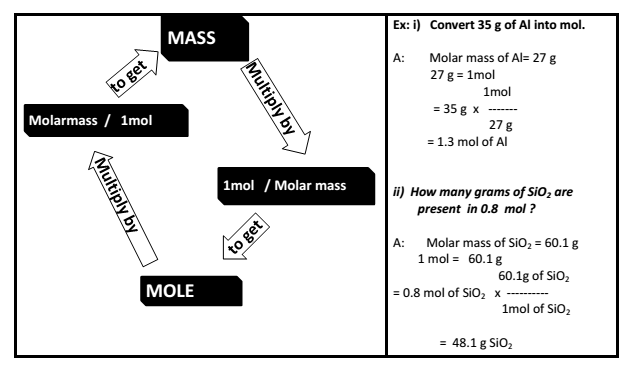
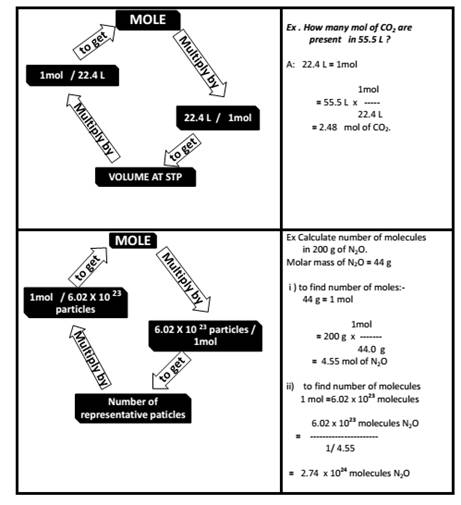
Atoms and Molecules class 9 Notes
- CBSE Revision notes (PDF Download) Free
- CBSE Revision notes for Class 9 Science PDF
- CBSE Revision notes Class 9 Science – CBSE
- CBSE Revisions notes and Key Points Class 9 Science
- Summary of the NCERT books all chapters in Science class 9
- Short notes for CBSE class 9th Science
- Key notes and chapter summary of Science class 9
- Quick revision notes for CBSE exams
CBSE Class-9 Revision Notes and Key Points
Atoms and Molecules class 9 Notes. CBSE quick revision note for Class-9 Science, Chemistry, Maths, Biology and other subject are very helpful to revise the whole syllabus during exam days. The revision notes covers all important formulas and concepts given in the chapter. Even if you wish to have an overview of a chapter, quick revision notes are here to do if for you. These notes will certainly save your time during stressful exam days.
- Revision Notes for class-09 Mathematics
- Revision Notes for class-09 Science
- Revision Notes for class-09 Social Science
- Revision Notes for class-09 English Communicative
To download Atoms and Molecules class 9 Notes, sample paper for class 9 Mathematics, Social Science, Science, English Communicative; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
- Matter in Our Surrounding class 9 Notes Science
- Is Matter around Us Pure class 9 Notes Science
- Atoms and Molecules class 9 Notes Science
- Structure of the Atoms class 9 Notes Science
- The Fundamental Unit of Life class 9 Notes Science
- Tissues class 9 Notes Science
- Diversity in Living Organisms class 9 Notes Science
- Motion class 9 Notes Science
- Forces and Laws of Motion class 9 Notes Science
- Gravitation class 9 Notes Science
- Work and Energy class 9 Notes Science
- Sound class 9 Notes Science
- Why Do We Fall ill class 9 Notes Science
- Natural Resources class 9 Notes Science
- Improvement in Food Resources class 9 Notes Science

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now