Amines class 12 Notes Chemistry in PDF are available for free download in myCBSEguide mobile app. The best app for CBSE students now provides Amines class 12 Notes latest chapter wise notes for quick preparation of CBSE board exams and school-based annual examinations. Class 12 Chemistry notes on chapter 13 Amines are also available for download in CBSE Guide website.
Class 12 Chemistry notes Chapter 13 Amines
Download CBSE class 12th revision notes for chapter 13 Amines in PDF format for free. Download revision notes for Amines class 12 Notes and score high in exams. These are the Amines class 12 Notes prepared by team of expert teachers. The revision notes help you revise the whole chapter 13 in minutes. Revision notes in exam days is one of the best tips recommended by teachers during exam days.
Download Revision Notes as PDF
CBSE Class 12 Chemistry Revision Notes Chapter 13 Amines
- Amines: Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group.
- Classification of amines:
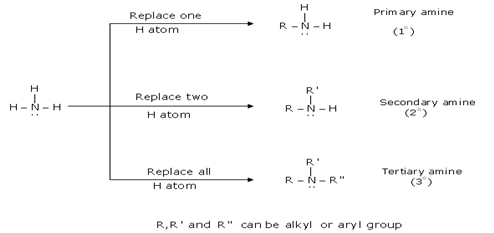
- Preparation of amines:
(i) By reduction of nitro compounds: Nitro compounds can be catalytically reduced by passing hydrogen gas in presence of Raney Ni, finely divided Pt or Pd as catalyst at room temperature.
a)
b)
Nitro compounds can also be reduced with active metals such as Fe, Sn, Zn etc. with conc. HCl.
a)
b)
(ii) By Hoffmann’s method (Ammonolysis of alkyl halides): Reaction of alkyl halides with an ethanolic solution of ammonia in a sealed tube at 373 K forms a mixture of primary, secondary and tertiary amine and finally quarternary ammonium salt. Process of cleavage of C-X bond by ammonia is called ammonolysis.
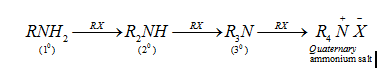
- The free amine can be obtained from the ammonium salt by treatment with a strong base:
a)
b)
c) - Order of reactivity of halides is: RI>RBr>RCl
- Larger the size of halogen atom easier is the cleavage of R-X bond
- Limitations of Hoffmann’s method: Method gives mixture of amines which are difficult to separate in a laboratory.
- Methods to get only one product by Hoffmann’s method:
(i) When ammonia is taken in excess primary amine is formed as main product
(ii) When alkyl halide is used in excess quarternary ammonium salt is formed as main product.
Method is not suitable for preparation of aryl amines because aryl amines are relatively less reactive than alkyl halides towards nucleophilic substitution reactions.
(iii) By reduction of nitriles: Nitriles can be reduced to amines using H2 / Ni , LiAlH4 or Na(Hg) / C2H5 OH
(iv) By reduction of amides: Amides are reduced to corresponding amines by LiAlH4
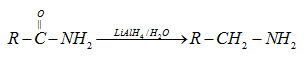
(v) By Gabriel phthalimide synthesis: Gabriel synthesis is used for the preparation of primary amines. When phthalimide is treated with ethanolic potassium hydroxide, it forms potassium salt of phthalimide which on heating further with alkyl halide followed by alkaline hydrolysis produces the corresponding primary amine.
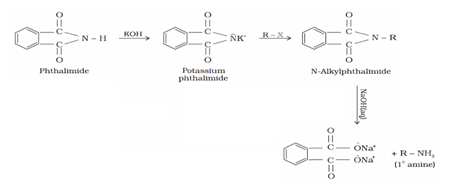
Aromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with potassium phthalimide.
(vi) By Hoffmann bromamide degradation reaction: Primary amines can be prepared from amides by treatment with Br2 and KOH. Amine contains one carbon atom less than the parent amide.
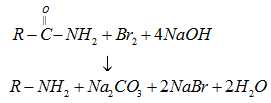
- Physical properties of amines:
(i) Solubility: Lower aliphatic amine is soluble in water because they can form hydrogen bonding with water. Solubility decreases with increases in molar mass of amines due to increase in size of hydrophobic group
(ii) Boiling points: Among the isomeric amines primary and secondary amines have high boiling point because they can form hydrogen bonding. Tertiary amine cannot form hydrogen bonding due to the absence of hydrogen atom available for hydrogen bond formation. Hence order of boiling of isomeric amines is Primary>Secondary> Tertiary
- Chemical properties of amines:
(a) Basic character of amines: Amines have an unshared pair of electrons on nitrogen atom due to which they behave as Lewis base. Basic character of amines can be better understood in terms of their Kb and pKb values
Or
Greater Kb value or smaller pKb indicates base is strong.
(b) Comparison of basic strength of aliphatic amines and ammonia: Aliphatic amines are stronger bases than ammonia due to +I effect of alkyl groups leading to high electron density on the nitrogen atom.
(c) Comparison of basic strength of primary, secondary and tertiary amines
(i) The order of basicity of amines in the gaseous phase follows the expected order on the basis of +I effect: tertiary amine > secondary amine > primary amine > NH3
(ii) In aqueous solution it is observed that tertiary amines are less basic than either primary or secondary amines. This can be explained on basis of following factors:
a) Solvation effect: Greater is the stability of the substituted ammonium cation formed, stronger is the corresponding amine as a base. Tertiary ammonium ion is less hydrated than secondary ammonium ion which is less hydrated than primary amine. Thus tertiary amines have fewer tendencies to form ammonium ion and consequently are least basic.
On the basis of solvation effect order of basicity of aliphatic amines should be primary amine>secondary amine>tertiary amine.
b) Steric factor: As the crowding of alkyl group increases from primary to tertiary amine hinderance to hydrogen bonding increases which eventually decreases the basic strength. Thus there is a subtle interplay of the inductive effect, solvation effect and steric hinderance of the alkyl group which decides the basic strength of alkyl amines in the aqueous state.
When the alkyl group is small like CH3 there is no steric hindrance to hydrogen bonding. In this case order of basicity in aqueous medium is
When alkyl group is ethyl group order of basicity in aqueous medium is
c) Comparison of basic strength of aryl amines and alkylamines: Generally aryl amines are considerably less basic than alkyl amines .Taking an example of aniline and ethylamine it is observed that ethyl amine is more basic than aniline. In aniline –NH2 group is directly attached to benzene ring. Hence unshared pair of electron on nitrogen is less available for protonation because of resonance. Below mentioned are resonating structures of aniline.
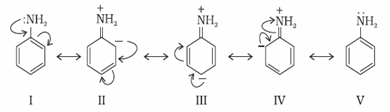
In the above resonating structures there is a positive charge on nitrogen atom making the lone pair less available for protonation. Hence aniline is less basic than ethyl amine which has no resonating structures. Less basicity of aniline can also be explained by comparing the relative stability of aniline and anilinium ion obtained by accepting a proton. Greater the number of resonating structures, greater is the stability of that species.
Aniline is resonance hybrid of five resonating structures whereas anilinium ion has only two resonating structures.
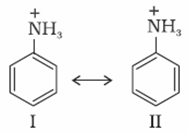
Thus aniline has less tendency to accept a proton to form anilinium ion.
d) Effect of substituent on basic character of amines: Electron donating or electron releasing group/groups (EDG) increases basic strength while electron withdrawing (EWG) decreases basic strength.
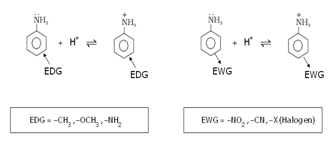
- Reactions of amines:
a) Acylation Reaction: Aliphatic and aromatic primary and secondary amines (which contain replaceable hydrogen atoms) react with acid chlorides, anhydrides and esters to form substituted amide. Process of introducing an acyl group (R-CO-) into the molecule is called acylation. The reaction is carried out in the presence of a stronger base than the amine, like pyridine, which removes HCl formed and shifts the equilibrium to the product side.

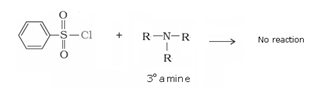
Since tertiary amine do not contain replaceable hydrogen atom they do not undergo acylation reaction.
b) Carbylamine reaction: Only aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form isocyanides or carbylamines.
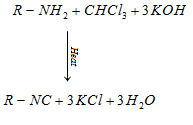
Secondary and tertiary amines do not give the above test.
c) Reaction of primary amine with nitrous acid:
(i) Primary aliphatic amine on reaction with nitrous acid (HNO2) forms aliphatic diazoniumsalt which decomposes to form alcohol and evolve nitrogen.
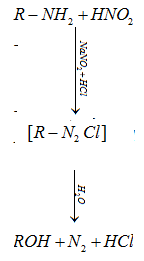
(ii) Primary aromatic amines react with nitrous acid (HNO2) in cold (273-278 K) to form diazonium salt.
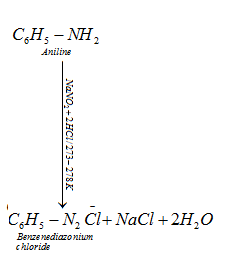
d) Reaction with benzene sulphonyl chloride: Hinsberg’s reagent-Benzenesulphonyl chloride (C6H5SO2Cl) reacts with primary and secondary amines to form sulphonamides.
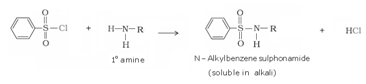
The hydrogen attached to nitrogen in sulphonamide formed by primary amine is strongly acidic due to the presence of strong electron withdrawing sulphonyl group. Hence, it is soluble in alkali.
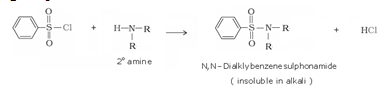
Since sulphonamide formed by secondary amine does not contain any hydrogen atom attached to nitrogen atom, so it is not acidic. Hence it is insoluble in alkali.
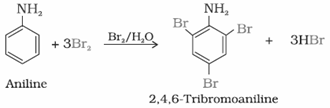
- Ring substitution in aromatic amine: Aniline is more reactive than benzeneand undergoes electrophilic substitution reaction preferably at ortho and para position.
(i) Bromination: Aniline reacts with bromine water at room temperature to give a white precipitate of 2, 4, 6-tribromoaniline
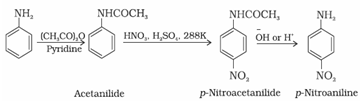
In order to stop reaction at monosubstitution activating effect of –NH2 group is reduced by acetylation. This prevents di and tri substituted products. Acetyl group is removed by hydrolysis.
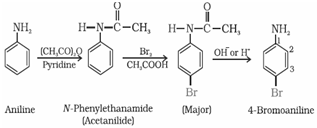
(ii) Nitration:
(a) Under strongly acidic medium aniline gets protonated to form anilinium ion, which is deactivating group and is meta directing. Hence minitroaniline is also formed in 47% along with ortho and para products.
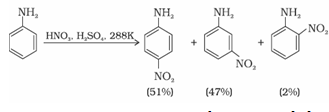
Aromatic amines cannot be nitrated directly because HNO3 being a strong oxidising agent oxidises it forming black mass.
(b) Nitration by protecting the –NH2 group by acetylation reaction with acetic anhydride:
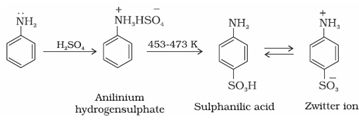
iii) Sulphonation: Aniline reacts with conc. H2SO4 to form aniliniumhydrogensulphate which on heating with sulphuric acid at 453-473K produces p-aminobenzenesulphonic acid, commonly known as sulphanilic acid, as the major product.
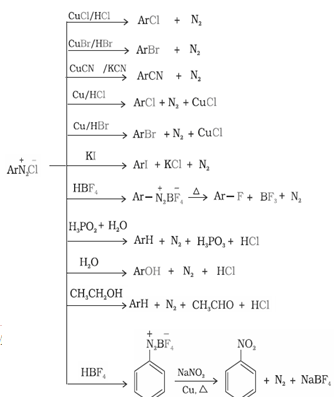
- Reactions of benzene diazonium chloride:
a) Reactions involving displacement of nitrogen:
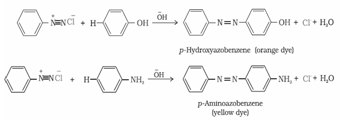
b) Reactions involving retention of diazo group, coupling reactions: Diazonium ion acts as an electrophile because there is a positive charge on terminal nitrogen. Therefore benzene diazonium chloride couples with electron rich compounds like phenol and aniline to give azo compounds. Azo compounds contain –N=N- bond and reaction is coupling reaction.
CBSE Class 12 Revision Notes and Key Points
Amines class 12 Notes Chemistry. CBSE quick revision note for class-12 Chemistry, Physics Math’s, Biology and other subject are very helpful to revise the whole syllabus during exam days. The revision notes covers all important formulas and concepts given in the chapter. Even if you wish to have an overview of a chapter, quick revision notes are here to do if for you. These notes will certainly save your time during stressful exam days.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Economics
- Business Studies
- Computer Science
- Informatics Practices
- English Core
- History
- Physical Education
To download Amines class 12 Notes Chemistry, sample paper for class 12 Physics, Chemistry, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies, and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT Amines, NCERT Exemplar Amines, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
- Solid State class 12 Notes Chemistry
- Solutions class 12 Notes Chemistry
- Electrochemistry class 12 Notes Chemistry
- Chemical Kinetics class 12 Notes Chemistry
- Surface Chemistry class 12 Notes Chemistry
- General Principles and Processes of Isolation of Elements class 12 Notes Chemistry
- The p-Block Elements class 12 Notes Chemistry
- The d- and f- Block Elements class 12 Notes Chemistry
- Coordination Compounds class 12 Notes Chemistry
- Haloalkanes and Haloarenes class 12 Notes Chemistry
- Alcohols Phenols and Ethers class 12 Notes Chemistry
- Aldehydes Ketones and Carboxylic Acids class 12 Notes Chemistry
- Amines class 12 Notes Chemistry
- Biomolecules class 12 Notes Chemistry
- Polymers class 12 Notes Chemistry
- Chemistry in Everyday Life class 12 Notes Chemistry

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now