NCERT Solutions class 12 Chemistry The Solid State part 2 Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for Chemistry as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER ONE THE SOLID STATE
- 1.1 General Characteristics of Solid State
- 1.2 Amorphous and Crystalline Solids
- 1.3 Classification of Crystalline Solids
- 1.4 Crystal Lattices and Unit Cells
- 1.5 Number of Atoms in a Unit Cell
- 1.6 Close-Packed Structure
- 1.7 Packing Efficiency
- 1.8 Calculations Involving Unit Cell Dimensions
- 1.9 Imperfections in Solids
- 1.10 Electrical Properties
- 1.11 Magnetic Properties
NCERT Solutions class 12 Chemistry The Solid State part 2
14. If the radius of the octahedral void is r and radius of the atoms in close packing is R, derive relation between r and R.
15. Copper crystallises into a fcc lattice with edge length 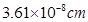 . Show that the calculated density is in agreement with its measured value of 8.92 g
. Show that the calculated density is in agreement with its measured value of 8.92 g 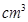 .
.
16. Analysis shows that nickel oxide has the formula 0.98O1.00. What fractions of nickel exist as 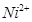 and
and 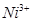 ions?
ions?
17. What is a semiconductor? Describe the two main types of semiconductors and contrast their conduction mechanism.
18. Non-stoichiometric cuprous oxide, 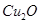 can be prepared in laboratory. In this oxide, copper to oxygen ratio is slightly less than 2:1. Can you account for the fact that this substance is a p-type semiconductor
can be prepared in laboratory. In this oxide, copper to oxygen ratio is slightly less than 2:1. Can you account for the fact that this substance is a p-type semiconductor
19. Ferric oxide crystallises in a hexagonal close-packed array of oxide ions with two out of every three octahedral holes occupied by ferric ions. Derive the formula of the ferric oxide.
20. Classify each of the following as being either a p-type or an n-type semiconductor:
(i) Ge doped with In
(ii) B doped with Si.
21. Gold (atomic radius = 0.144 nm) crystallises in a face-centred unit cell. What is the length of a side of the cell?
22. In terms of band theory, what is the difference
(i) Between a conductor and an insulator
(ii) Between a conductor and a semiconductor
23. Explain the following terms with suitable examples:
(i) Schottky defect
(ii) Frenkel defect
(iii) Interstitials and
(iv) F-centres
24. Aluminium crystallises in a cubic close-packed structure. Its metallic radius is 125 pm.
(i) What is the length of the side of the unit cell?
(ii) How many unit cells are there in 1.00 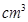 of aluminium?
of aluminium?
25. If NaCl is doped with 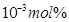 of
of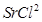 , what is the concentration of cation vacancies?
, what is the concentration of cation vacancies?
26. Explain the following with suitable examples:
(i) Ferromagnetism
(ii) Paramagnetism
(iii) Ferrimagnetism
(iv) Antiferromagnetism
(v) 12-16 and 13-15 group compounds.
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
NCERT Solutions for Class 12 Chemistry
Class 12 Chemistry NCERT book Solutions covers both organic and inorganic chemistry. Please note that CBSE has reduced the syllabus for class 12 Chemistry. So, you must check the latest syllabus of CBSE while going through the NCERT solution of any chapter given in the class 12 NCERT book.
- The Solid State part-1
- The Solid State part-2
- Solutions In-text question
- Solutions Part 1
- Solutions Part 2
- Solutions In-text question
- Electrochemistry In-text question
- Electrochemistry part 1
- Electrochemistry part 2
- Chemical kinetics In-text question
- Chemical kinetics part 1
- Chemical kinetics part 2
- Surface Chemistry In-text Questions
- Surface Chemistry part 1
- Surface Chemistry part 2
- General principle and processes of isolation of elements In-text Questions
- General principle and processes of isolation of elements part-1
- General principle and processes of isolation of elements part-2
- The p block elements part 1
- The p block elements part 2
- The p block elements In-text Questions
- The d and f block elements In-text Questions
- The d and f block elements part 1
- The d and f block elements part 2
- Coordination Compounds In-text Questions
- Coordination Compounds part 1
- Coordination Compounds part 2
- Haloalkanes and haloarenes In-text Questions
- Haloalkanes and haloarenes part 1
- Haloalkanes and haloarenes part 2
- Alcohol phenol and ether In-text Questions
- Alcohol phenol and ether part 1
- Alcohol phenol and ether part 2
- Aldehydes ketones and carboxylic acid In-text Questions
- Aldehydes ketones and carboxylic acid part 1
- Aldehydes ketones and carboxylic acid part 2
- Amines In-text Questions
- Amines part 1
- Amines part 2
- Biomolecules In-text Questions
- Biomolecules part 1
- Biomolecules part 2
- Polymers In-text Questions
- Polymers part 1
- Polymers part 2
- Chemistry in everyday life In-text Questions
- Chemistry in everyday life part 1
- Chemistry in everyday life part 2

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now