NCERT Solutions class-11 Chemistry Equilibrium Part-2
26. Which of the following reactions will get affected by increasing the pressure?
Also, mention whether change will cause the reaction to go into forward or backward direction.
(i) 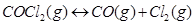
(ii) 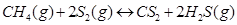
(iii) 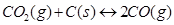
(iv) 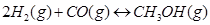
(v) 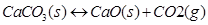
(vi) 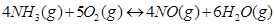
27. The equilibrium constant for the following reaction is 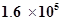 at 1024 K.
at 1024 K.
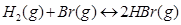
Find the equilibrium pressure of all gases if 10.0 bar of HBr is introduced into a sealed container at 1024 K.
28. Dihydrogen gas is obtained from natural gas by partial oxidation with steam as per following endothermic reaction:
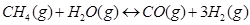
(a) Write as expression for 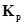 for the above reaction.
for the above reaction.
(b) How will the values of 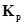 and composition of equilibrium mixture be affected by
and composition of equilibrium mixture be affected by
(i) Increasing the pressure
(ii) Increasing the temperature
(iii) Using a catalyst?
29. Describe the effect of:
a) Addition of 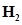
b) Addition of 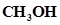
c) Removal of CO
d) Removal of 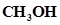
on the equilibrium of the reaction:
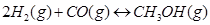
30. At 473 K, equilibrium constant 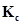 for decomposition of phosphorus pentachloride,
for decomposition of phosphorus pentachloride,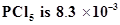 . If decomposition is depicted as,
. If decomposition is depicted as, 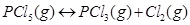
a) Write an expression for 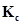 for the reaction.
for the reaction.
b) What is the value of 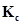 for the reverse reaction at the same temperature?
for the reverse reaction at the same temperature?
c) What would be the effect on 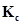 if (i) more
if (i) more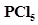 is added (ii) pressure is increased? (iii) The temperature is increased?
is added (ii) pressure is increased? (iii) The temperature is increased?
31. Dihydrogen gas used in Haber’s process is produced by reacting methane from natural gas with high temperature steam. The first stage of two stage reaction involves the formation of CO and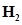 . In second stage, CO formed in first stage is reacted with more steam in water gas shift reaction,
. In second stage, CO formed in first stage is reacted with more steam in water gas shift reaction,
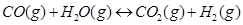
If a reaction vessel at 400°C is charged with an equimolar mixture of CO and steam such that 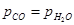 4.0 bar, what will be the partial pressure of
4.0 bar, what will be the partial pressure of 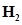 at equilibrium?
at equilibrium? 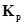 = 10.1 at 400°C
= 10.1 at 400°C
32. Predict which of the following reaction will have appreciable concentration of reactants and products:
a) 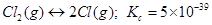
b) 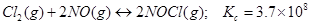
c) 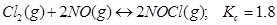
33. The value of 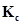 for the reaction
for the reaction 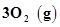
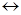
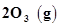 is
is 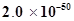 at 25°C. If the equilibrium concentration of O2 in air at 25°C is 1.6
at 25°C. If the equilibrium concentration of O2 in air at 25°C is 1.6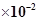 , what is the concentration of
, what is the concentration of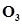 ?
?
34. The reaction, CO(g) + 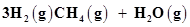 is at equilibrium at 1300 K in a 1L flask. It also contain 0.30 mol of CO, 0.10 mol of
is at equilibrium at 1300 K in a 1L flask. It also contain 0.30 mol of CO, 0.10 mol of 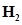 and 0.02 mol of
and 0.02 mol of 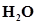 and an unknown amount of
and an unknown amount of 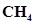 in the flask. Determine the concentration of
in the flask. Determine the concentration of 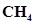 in the mixture. The equilibrium constant,
in the mixture. The equilibrium constant, 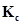 for the reaction at the given temperature is 3.90.
for the reaction at the given temperature is 3.90.
35. What is meant by the conjugate acid-base pair? Find the conjugate acid/base for the following species: 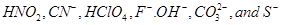
36. Which of the followings are Lewis acids?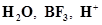 , and
, and 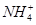
37. What will be the conjugate bases for the Bronsted acids: HF,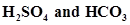 ?
?
38. Write the conjugate acids for the following Bronsted bases: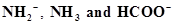 .
.
39. The species: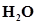 ,
,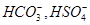 , and
, and 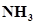 can act both as Bronsted acids and bases. For each case give the corresponding conjugate acid and base.
can act both as Bronsted acids and bases. For each case give the corresponding conjugate acid and base.
40. Classify the following species into Lewis acids and Lewis bases and show how these act as Lewis acid/base: (a)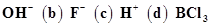 .
.
41. The concentration of hydrogen ion in a sample of soft drink is 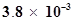 M. what is its pH?
M. what is its pH?
42. The pH of a sample of vinegar is 3.76. Calculate the concentration of hydrogen ion in it.
43. The ionization constant of HF, HCOOH and HCN at 298K are 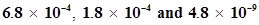 respectively. Calculate the ionization constants of the corresponding conjugate base.
respectively. Calculate the ionization constants of the corresponding conjugate base.
44. The ionization constant of phenol is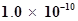 . What is the concentration of phenolate ion in 0.05 M solution of phenol? What will be its degree of ionization if the solution is also 0.01M in sodium phenolate?
. What is the concentration of phenolate ion in 0.05 M solution of phenol? What will be its degree of ionization if the solution is also 0.01M in sodium phenolate?
45. The first ionization constant of 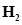 S is
S is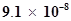 . Calculate the concentration of HS- ion in its 0.1 M solution. How will this concentration be affected if the solution is 0.1 M in HCl also? If the second dissociation constant of
. Calculate the concentration of HS- ion in its 0.1 M solution. How will this concentration be affected if the solution is 0.1 M in HCl also? If the second dissociation constant of 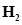 S is
S is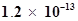 , calculate the concentration of
, calculate the concentration of 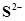 under both conditions.
under both conditions.
46. The ionization constant of acetic acid is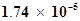 . Calculate the degree of dissociation of acetic acid in its 0.05 M solution. Calculate the concentration of acetate ion in the solution and its pH.
. Calculate the degree of dissociation of acetic acid in its 0.05 M solution. Calculate the concentration of acetate ion in the solution and its pH.
47. It has been found that the pH of a 0.01M solution of an organic acid is 4.15. Calculate the concentration of the anion, the ionization constant of the acid and its pKa.
48. Assuming complete dissociation, calculate the pH of the following solutions:
(a) 0.003 M HCl
(b) 0.005 M NaOH
(c) 0.002 M HBr
(d) 0.002 M KOH
49. Calculate the pH of the following solutions:
a) 2 g of TlOH dissolved in water to give 2 litre of solution.
b) 0.3 g of 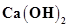 dissolved in water to give 500 mL of solution.
dissolved in water to give 500 mL of solution.
c) 0.3 g of NaOH dissolved in water to give 200 mL of solution.
d) 1mL of 13.6 M HCl is diluted with water to give 1 litre of solution.
50. The degree of ionization of a 0.1M bromoacetic acid solution is 0.132. Calculate the pH of the solution and the 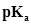 of bromoacetic acid.
of bromoacetic acid.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now