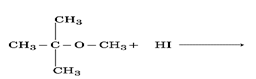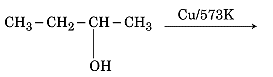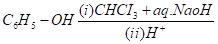CBSE Question Paper 2016 class 12 Chemistry conducted by Central Board of Secondary Education, New Delhi in the month of March 2016. CBSE previous year question papers with the solution are available in the myCBSEguide mobile app and website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
CBSE Question Paper 2016 class 12 Chemistry

Class 12 Chemistry list of chapters
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d and f Block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday life
CBSE Question Paper 2016 Class 12 Chemistry
General Instructions:
- All questions are compulsory.
- Questions number 1 to 5 are very short answer questions and carry 1 mark each.
- Questions number 6 to 10 are short answer questions and carry 2 marks each.
- Questions number 11 to 22 are also short answer questions and carry 3 marks each.
- Question number 23 is a value based question and carry 4 marks.
- Questions number 24 to 26 are long answer questions and carry 5 marks each.
- Use log tables, if necessary. Use of calculators is not allowed.
- Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction.
- Pb(NO3)2 on heating gives a brown gas which undergoes dimerization on cooling? Identify the gas.
- Give an example each of a molecular solid and an ionic solid.
- Write the IUPAC name of the given compound:
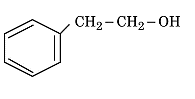
- What is the reason for the stability of colloidal sols?
- Gas (A) is more soluble in water than Gas (B) at the same temperature.
Which one of the two gases will have the higher value of KH (Henry’s constant) and why? - In non-ideal solution, what type of deviation shows the formation of maximum boiling azeotropes ?
- Gas (A) is more soluble in water than Gas (B) at the same temperature.
- Write the structures of the following:
- BrF3
- XeF4
OR
What happens when:
- SO2 gas is passed through an aqueous solution Fe3+salt?
- XeF4 reacts with SbF5?
- When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3 moles of AgCl are precipitated per mole of the compound. Write
- Structural formula of the complex
- IUPAC name of the complex
- For reaction: H2+CI2
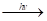 2HCI
2HCI
Rate=K- Write the order and molecularity of this reaction.
- Write the unit of k.
- Write the chemical equations involved in the following reactions:
- Hoffmann-bromamide degradation reaction
- Carbylamine reaction
- An element crystallizes in a b.c.c. lattice with cell edge of 500pm. The density of the element is 7.5g cm^-^3cm^-^3. How many atoms are present in 300 g of the element?
- For the first order thermal decomposition reaction, the following data were obtained:
C2H5Cl(g)→C2H4(g)+HCl(g)Time / sec Total pressure / atm 0 0.30 300 0.50 Calculate the rate constant
(Given: log 2=0.301, log 3=0.4771, log 4=0.6021) - Define the following terms:
- Lyophilic colloid
- Zeta potential
- Associated colloids
- Name the method of refining of nickel
- What is the role of cryolite in the extraction of aluminium?
- What is the role of limestone in the extraction of iron from its oxides?
- Calculate the boiling point of solution when 4 g of MgSO4 (M=120 g mol-1) was dissolved in 100 g of water, assuming MgSO4 undergoes complete ionization. (Kb for water=0.52 K kg mol-1)
- Give reasons:
- SO2 is reducing while TeO2 is an oxidizing agent.
- Nitrogen does not form pentahalide.
- ICl is more reactive than I2.
- Write the final product (s) in each of the following reactions:
- Give reasons for the following:
- Aniline does not undergo Friedal-Crafts reaction.
- (CH3)2 NH is more basic than (CH3)3N in an aqueous solution.
- Primary amines have higher boiling point than tertiary amines.
- How do you convert:
OR
Write the major product(s) in the following:
- Chlorobenzene to biphenyl
- Propene to 1-iodopropane
- 2-bromobutane to but-2-ene
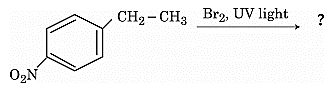
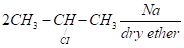
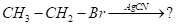
- What is the role of Sulphur in the vulcanization of rubber?
- Identify the monomers in the following polymer:
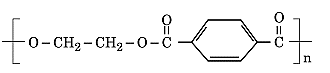
- Arrange the following polymers in the increasing order of their intermolecular forces:
Terylene, Polythene, Neoprene
- Write the structural difference between starch and cellulose.
- What type of linkage is present in Nucleic acids?
- Give one example each for fibrous protein and globular protein.
- For the complex [Fe(H2O)6]3+, write the hybridization, magnetic character and spin of the complex. (At. number: Fe=26)
- Draw one of the geometrical isomers of the complex [Pt(en)2CI2]2+ which is optically inactive.
- Due to hectic and busy schedule, Mr. Singh started taking junk food in the lunch break and slowly became habitual of eating food irregularly to excel in his field. One day during meeting he felt severe chest pain and fell down. Mr. Khanna, a close friend of Mr. Singh. took him to doctor immediately. The doctor diagnosed that Mr. Singh was suffering from acidity and prescribed some medicines. Mr. Khanna advised him to eat home made food and change his lifestyle by doing yoga, meditation and some physical exercise. Mr. Singh followed his friend’s advice and after few days he started feeling better. After reading the above passage, answer the following:
- What are the values (at least two) displayed by Mr. Khanna?
- What are antacids? Give one example.
- Would it be advisable to take antacids for a long period of time? Give reason.
- Calculate E˚cell for the following reaction at 298K:
2Al(s)1 3Cu2+(0.01M)→2Al3+ (0.01M) 1 3Cu(s)
Given: Ecell = 1.98 V - Using the E˚ values of A and B, predict which is better for coating the surface of iron [E°(Fe2+/Fe)=-0.44V] to prevent corrosion and why ?
Given: E˚(A2+/A)=-2.37V : E° (B2+/B)=-0.14V
-
OR
- The conductivity of 0.001 molL-1 solution of CH3COOH is 3.905X 10-5S cm-1 Calculate its molar conductivity and degree of dissociation (∝).
Given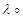 (H+)=349.6 S cm2 mol-1 and
(H+)=349.6 S cm2 mol-1 and 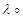 (CH3COO–)=40.9 S cm2 mol-1
(CH3COO–)=40.9 S cm2 mol-1 - Define electrochemical cell. What happens if external potential applied becomes greater than E°Cell of electrochemical cell?
- The conductivity of 0.001 molL-1 solution of CH3COOH is 3.905X 10-5S cm-1 Calculate its molar conductivity and degree of dissociation (∝).
-
- Account for the following:
- Mn shows the highest oxidation state of +7 with oxygen but with fluorine it shows the highest oxidation state of +4.
- Cr2+ is a strong reducing agent.
- Cu2+ salts are coloured while Zn2+ salts are white.
- Complete the following equations:
- 2MnO2+4KOH+O2
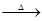
- CrO
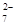 +14H++61-→→
+14H++61-→→
- 2MnO2+4KOH+O2
OR
- The elements of 3d transition series are given as:
Sc Ti V Cr Mn Fe Co Ni Cu Zn - Answer the following:
- Write the element which shows maximum number of oxidation states.
Give reason. - Which element has the highest m.p?
- Which element shows only +3 oxidation state?
- Which element is a strong oxidizing agent in +3 oxidation state and why?
- Write the element which shows maximum number of oxidation states.
- Account for the following:
-
- Write the structures of A and B in the following reactions:
- CH3COCl
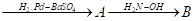
- CH3MgBr
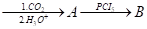
- CH3COCl
- Distinguish between:
- C6H5-COCH3 and C6H5-CHO
- CH3COOH and HCOOH
- Arrange the following in the increasing order of their boiling points:
CH3CHO, CH3COOH, CH3C2OH
OR
- Write the chemical reaction involved in Wolff-Kishner reduction.
- Arrange the following in the increasing order of their reactivity towards nucleophilic addition reaction:
C6H5COCH3, CH3-CHO, CH3COCH3 - Why carboxylic acid does not give reactions of carbonyl group?
- Write the product in the following reaction
CH3CH2CH=CH2CH2CN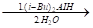
- A and B are two functional isomers of compound C3H6O. On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
- Write the structures of A and B in the following reactions:
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
Chemistry Question Paper 2016
Download class 12 Chemistry question paper with solution from best CBSE App the myCBSEguide. CBSE class 12 Chemistry question paper 2016 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper. CBSE question papers 2016 for class 12 Chemistry have 19 questions with solution.
Previous Year Question Paper for class 12 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 209, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Business Studies
- Economics
- History
- Geography
- Political Science
- Physical Education
- Computer Science
- Informatics Practices
- English Core
- Hindi Core
- Hindi Elective
- Other Subjects
To download CBSE Question Paper 2016 class 12 Accountancy, Chemistry, Physics, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now