Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Rakesh Pateer 5 years, 7 months ago
- 2 answers
Posted by Edwin Hmar Keivawm 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
Features of the Harappan culture:
1. Urban Civilisation: The sight of the towns of Mohenjodaro and Harappa reflect the supremacy of the then architecture and town planning. There were wide roads ranging from 9 to 34 feet in width. The horizontal roads cut the vertical ones on right angles. The towns had brick houses, both big and small ones. There was provision of cross ventilation and sufficient lighting. The sight of drains and huge dustbins outside the houses revealed that there must have been some local institution to manage sanitation and such other activities smoothly.
2. Architecture: There were varieties of building that were unearthed:
(i) Buildings for the dwelling of the citizens.
(ii) Buildings for public purposes.
(iii) Public fire places.
3. The Great Bath: The Great Bath, discovered during excavations was 39 feet in length, 23 feet in width and 8 feet in depth.
Verandahs and rooms were constructed around it. There was a provision of filling the bath with water and emptying it. There was a well closely which might have been serving as the source of filling fresh water into the bath. This structure is an excellent symbol of the Indus Valley Civilisation.
4. Food: The people of Indus Valley ate barley, wheat, fruit and flesh also.
5. Clothes: The available needles and spinning and weaving tools and button show that the people knew spinning and weaving. Both the cotton and woollen clothes were worn. The people wore coloured clothes. There was a close similarity in the clothes of men and women-folk.
6. Ornaments: Both the men and the women wore ornaments. The chief ornaments used to be armlets, necklace, ear-rings, metal ring (bangle), belt etc.
7. Cosmetics: Hair used to be decorated in many forms. Brass combs, mirrors, piece of decoration made of ivory and a type of colour used to redden the lips like lipstick too have been found.
8. Amusements: The people used to utilise their time in hunting, playing chess, music, dance, drawing and painting, taming birds and catching fish. These formed the sources of their recreation.
9. Art and craft: Patterns of fine drawing are available on the seals of the times. The clay carts and toys too are fine specimen of the grand art.
10. Religion: There are various hypothesis regarding religion but we still do not know, there are indication that they could have worshipped nature, mother goddess, proto shiva etc.
Posted by Salman Wazir 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
Tournament is a large contest of many rounds among various teams. It is a competition held among various teams in a particular activity according to a fixed schedule where a winner is decided.
Number of teams participating are 27.
Number of matches = n – 1 = 27 – 1 = 26 matches.
Teams in upper half = n+1/2=27+1/2= 28/2
Teams in lower half = n−1/2=27−1/2= 26/2 = 13 teams
Number of teams participating are odd so number of byes will be = 32 – 27 = 5 byes.
In Knockout system the team which loses the match is eliminated and the winners continue to play. When we have less time usually we conduct single elimination tournament.
Date/time and court number, is specified for each match so that teams/players report for their matches well in advance.
Posted by Dyavathi Rakesh 5 years, 7 months ago
- 2 answers
? Yang ? 5 years, 7 months ago
Gaurav Seth 5 years, 7 months ago
In a circuit the current flowing through the circuit is given as :
I=E/(r+R)
Where r is the internal resistance of the cell and R is the external resistance.
Maximum current can be only drawn when external resistance is zero.
Posted by Vandana Pandit 5 years, 7 months ago
- 1 answers
Posted by Vandana Pandit 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
Ans: Functional structure as the people are being grouped on the basis of functions
Explanation: Functional Structure: In functional structure activities are grouped and departments are created on the basis of specific functions to be performed. For example, all the jobs related to production are grouped under production department, sales departments etc.
Posted by Komal Wahla 5 years, 7 months ago
- 1 answers
Posted by Ishu Bansal 5 years, 7 months ago
- 0 answers
Posted by Muskan Jakhar 5 years, 7 months ago
- 2 answers
Posted by Sunil Sharma Sunil Sharma 5 years, 6 months ago
- 0 answers
Posted by Gaurav Singh 5 years, 7 months ago
- 2 answers
A Roy 5 years, 7 months ago
Gaurav Seth 5 years, 7 months ago
The year 1947 was the year of one of the largest, most abrupt, unplanned and tragic transfer of population which human history has known.
- There were killings and atrocities on both sides of the border. Cities such as Lahore, Amritsar and Kolkata (Calcutta) became divided into 'communal zones'. Muslims would avoid going into an area where mainly Hindus or Sikhs lived and vice versa.
- Minorities on both sides of the border fled their home and often secured temporary shelter in 'refugee camps'. They travelled to the other side of the new border by all sorts of means, often by foot. Even during this journey, they were often attacked, killed or raped. Thousands of women were abducted on both sides of the border. Often women were killed by their own family members to preserve the 'family honour'. Many children were separated from their parents.
- People who managed to cross the border did not have a place they could call home. For lakhs of these 'refugees', the country's freedom meant life in 'refugee camps' for months and sometimes for years.
- Partition was not merely a division of properties, liabilities and assets, or a political division of the country and the administrative apparatus. Also divided were things such as tables, chairs, typewriters, paper-clips and books.
- Even after large-scale migration of Muslims to the newly created Pakistan, the Muslim population in India accounted for 12% of the total population in 1951. The Partition had already created severe conflict between the two communities.
Above all, it was a violent separation of communities who had hitherto lived together as neighbours for centuries.
Posted by Pranav Kadyan 5 years, 7 months ago
- 3 answers
Aashi Chaurasia 5 years, 7 months ago
Shiva N.?? Yadav?? 5 years, 1 month ago
Posted by Ragini Yadav??? 5 years, 7 months ago
- 1 answers
Anshika Sharma 5 years, 7 months ago
Posted by Ragini Yadav??? 5 years, 7 months ago
- 1 answers
Posted by Ragini Yadav??? 5 years, 7 months ago
- 2 answers
Insha Khalid 5 years, 7 months ago
Riya R 5 years, 7 months ago
Posted by Nisha Yadav 5 years, 7 months ago
- 2 answers
Gaurav Seth 5 years, 7 months ago
Aminoacylation:It is a two stage process whereby amino acids are 'activated' in order to be incorporated into proteins; energy in the form of ATP is required and one of a family of 20 aminoacyl-tRNA synthetases, one specific for each amino acid, links the carboxyl group of the amino acid to either the 2′- or 3′-hydroxyl group of the ribose unit at the 3′-end of tRNA.
Amino acids are activated in the presence of ATP and linked to (cognate) t-RNA
Posted by Ragini Yadav??? 5 years, 7 months ago
- 2 answers
Gaurav Seth 5 years, 7 months ago
Henry law states whenever solid is dissolved in liquid or gas is dissolved in liquid its solubility is determined by pressure and temperature. it states that partial pressure is directly proportional to mole fraction. ie . p=khx where kh is Henry law constant. Raoult law states that when two volatile liquid are mixed their vapour pressure is directly proportional to mole fraction ie p1 = p*1 x
Posted by Nisha Yadav 5 years, 7 months ago
- 2 answers
Priyanshu Kumar 5 years, 7 months ago
Gaurav Seth 5 years, 7 months ago
a) If both the strands act as template, they would code for RNA molecule with different sequences, and in turn they code for different proteins.
b) the two RNA molecules if produced simultaneously would be complementary to each other , hence would for a double stranded RNA.
Posted by Mr. Badmash 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
The order from Berlin is called a thunderclap by Franz, because it was a complete shock for him. He had never thought that he would be deprived of the right of learning his native language. He realised he had so much more yet to learn and was going to lose the opportunity. When Franz reached the school, he saw a strange stillness in the class. Then the teacher, M. Hamel, announced that it was their last lesson in French and the new German teacher will take the charge on the following day. These words of M. Hamel came as thunderclap to Franz since the announcement was unexpected and sudden for him.
Posted by Aradhya Singh 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
Intermediate goods refer to those goods which are used either for resale or for further production in the same year. They are not ready for use in the sense some value has to be added to the intermediate goods. They are still within the production boundary.
two examples of intermediate goods
(i) Milk used in dairy shop for resale; (ii) Coal used In factory for further production.
Posted by Aditya Dadhich 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
Furniture purchased by a school is a final product because it is purchased for investment. School buy furniture for long - term use and it is considered as an investment. School is the final user of the furniture and no value is to be added to the furniture. This will be deemed as investment expenditure because furniture is used by the school for several years and is of high value.
Posted by Ankit Singh 5 years, 7 months ago
- 1 answers
Posted by Sukhjeet Kaur 5 years, 7 months ago
- 0 answers
Posted by Prathistha Thakur 5 years, 7 months ago
- 0 answers
Posted by Sandeep Singh 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
The faces of these children are pale and lifeless. They and their hair are like ‘rootless weeds’. The burden of fife makes them sit with their head ‘weighed down’. The stunted growth is depicted by ‘the paper-seeming bo/ and ‘the stunted unlucky heir of twisted bones’. Their weak bodies recite their fathers’ ‘gnarled disease’.
Posted by Komal Panwar 5 years, 7 months ago
- 1 answers
Gaurav Seth 5 years, 7 months ago
|
NOTICE National Public School 15 January 20XX Change in School Timings This is to inform all the students that there is a change in the school timings. From 20 January onwards, the school shall start from 9:00 a.m. instead of 8:00 a.m. The change is made due to the extreme cold weather and the fog in the morning. All the students are requested to adhere to the new timing.
Priyanka Jha (Principal) |
Posted by Abhi Jal 5 years, 7 months ago
- 0 answers
Posted by Harsh Tiwari 5 years, 7 months ago
- 2 answers
? Yang ? 5 years, 7 months ago
Posted by Divya Jyotsna 5 years, 7 months ago
- 0 answers

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide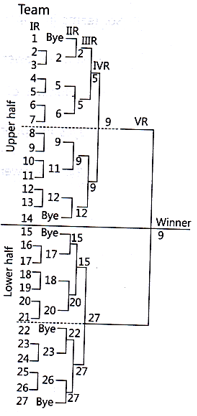

Lalit Singh 5 years, 7 months ago
0Thank You