Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Jashan Preet 4 years, 1 month ago
- 0 answers
Posted by Aditi 4 years, 1 month ago
- 0 answers
Posted by Najma Shaikh 4 years, 1 month ago
- 0 answers
Posted by Payal Jain 4 years, 1 month ago
- 0 answers
Posted by Coding Master 4 years, 1 month ago
- 0 answers
Posted by Abhishek Kourav 4 years, 1 month ago
- 0 answers
Posted by Syeda Sameena Fatima 4 years, 1 month ago
- 0 answers
Posted by Swati Nidhi 4 years, 1 month ago
- 0 answers
Posted by Abhishek Vishwakarma 4 years, 1 month ago
- 1 answers
Posted by Kanggong Tayeng. 4 years, 1 month ago
- 0 answers
Posted by Rahul Yadav 4 years, 1 month ago
- 1 answers
Rama Gaming 3 years, 10 months ago
Posted by Satyam Sharma 4 years, 1 month ago
- 1 answers
Posted by Harsh Vardhan 4 years, 1 month ago
- 1 answers
Posted by Mukesh Kumar 4 years, 1 month ago
- 3 answers
Arun Arunkumar 4 years, 1 month ago
Pooja Mohan 4 years, 1 month ago
Posted by Lakshay Rajpoot 4 years, 1 month ago
- 1 answers
Hari Krishna 4 years, 1 month ago
Posted by Anushka Anushka 4 years, 1 month ago
- 0 answers
Posted by Roshan Toppo 4 years, 1 month ago
- 0 answers
Posted by Ayushi Jain 4 years, 1 month ago
- 1 answers
Ifra Naaz 4 years, 1 month ago
Posted by Parth Makhija 3 years, 3 months ago
- 1 answers
Preeti Dabral 3 years, 3 months ago
- Energy of the ground state (n = 1) = – (ionization energy) = –13.6 eV
The wavelength of the incident radiation, {tex}\lambda{/tex} = 975 {tex}\mathop {\text{A}}\limits^{\text{o}} {/tex}
{tex}\therefore{/tex} The energy of the incident photon = hc/{tex}\lambda{/tex}
= {tex}\frac{6.63 \times 10^{-34} \times 3 \times 10^{8}}{975 \times 10^{-10} \times 1.6 \times 10^{-19}}{/tex} = 12.75 eV
Let electron is exerted to nth orbit,
{tex}\Rightarrow{/tex} 12.75 = 13.6 {tex}\left(\frac{1}{1^{2}}-\frac{1}{n^{2}}\right){/tex}
{tex}\Rightarrow{/tex} n = 4
The quantum transitions to the less excited states gives six possible lines as follows:
n = 4 : (4 {tex}\rightarrow{/tex} 3), (4 {tex}\rightarrow{/tex} 2), (4 {tex}\rightarrow{/tex} 1)
n = 3 : (3 {tex}\rightarrow{/tex} 2), (3 {tex}\rightarrow{/tex} 1)
n = 2 : (2 {tex}\rightarrow{/tex} 1)
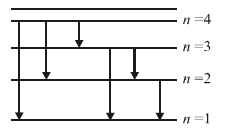
- The longest wavelength emitted is for the transitions (4 {tex}\rightarrow{/tex} 3) where energy difference is minimum.
Emin = (E4 - E3) = 13.6 {tex}\left(\frac{1}{3^{2}}-\frac{1}{4^{2}}\right){/tex} = 0.661 eV
Thus {tex}\lambda_{\max }=\frac{h c}{\mathrm{E}_{\min }}{/tex}
= {tex}\frac{6.63 \times 10^{-34} \times 3 \times 10^{8}}{0.661 \times 1.6 \times 10^{-19}} \mathrm{m}{/tex}
{tex}\approx{/tex} 18807 {tex}\mathop {\text{A}}\limits^{\text{o}} {/tex}
Posted by Bijay Roy 4 years, 1 month ago
- 1 answers
Posted by Alkesh Uikey 4 years, 1 month ago
- 1 answers
Tilok Das Sahu 2 years, 11 months ago
Posted by Lalit Mali 4 years, 1 month ago
- 2 answers
Shweta Sharma 4 years, 1 month ago
Riya Riya 4 years, 1 month ago
Posted by Chetna Surana 4 years, 1 month ago
- 0 answers
Posted by Bhumi Kumari 4 years, 1 month ago
- 0 answers

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide