
myCBSEguide App
Download the app to get CBSE Sample Papers 2023-24, NCERT Solutions (Revised), Most Important Questions, Previous Year Question Bank, Mock Tests, and Detailed Notes.
Install NowNCERT Solutions class 12 Chemistry The Solid State part 1 Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for Chemistry as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
NCERT Solutions class 12 Chemistry The Solid State part 1
1. Define the term ‘amorphous’. Give a few examples of amorphous solids.
2. What makes a glass different from a solid such as quartz? Under what conditions could quartz be converted into glass?
3. Classify the following as amorphous or crystalline solids:
Polyurethane, naphthalene, benzoic acid, teflon, potassium nitrate, cellophane, polyvinyl chloride, fibre glass, copper.
4. (i) What is meant by the term ‘coordination number’?
(ii) What is the coordination number of atoms:
(a) in a cubic close-packed structure?
(b) in a body-centred cubic structure?
5. How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
6. ‘Stability of a crystal is reflected in the magnitude of its melting point’. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules?
7. How will you distinguish between the following pairs of terms:
(i) Hexagonal close-packing and cubic close-packing?
(ii) Crystal lattice and unit cell?
(iii) Tetrahedral void and octahedral void?
8. How many lattice points are there in one unit cell of each of the following lattice?
(i) Face-centred cubic
(ii) Face-centred tetragonal
(iii) Body-centred
9. Explain
(i) The basis of similarities and differences between metallic and ionic crystals.
(ii) Ionic solids are hard and brittle.
10. Calculate the efficiency of packing in case of a metal crystal for
(i) simple cubic
(ii) body-centred cubic
(iii) face-centred cubic (with the assumptions that atoms are touching each other).
11. Silver crystallizes in fcc lattice. If edge length of the cell is 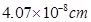 and density is 10.5 g
and density is 10.5 g 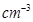 , calculate the atomic mass of silver.
, calculate the atomic mass of silver.
12. A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
13. Niobium crystallises in body–centred cubic structure. If density is 8.55 g 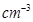 , calculate atomic radius of niobium using its atomic mass 93 u.
, calculate atomic radius of niobium using its atomic mass 93 u.
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
myCBSEguide
Question Bank, Mock Tests, Exam Papers, NCERT Solutions, Sample Papers, Notes
Install Now
Excellent for helping students