(i) used to determine heat changes
(ii) whose value is independent of path
(iii) used to determine pressure volume work
(iv) whose value depends on temperature only.
2. For the process to occur under adiabatic conditions, the correct condition is:
(i) ΔT = 0
(ii) Δp = 0
(iii) q = 0
(iv) w= 0
3. The enthalpies of all elements in their standard states are:
(i) unity
(ii) zero
(iii) < 0
(iv) different for each element
4. ΔU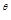 ¸of combustion of methane is – XkJ
¸of combustion of methane is – XkJ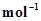 . The value of ΔH¸is
. The value of ΔH¸is
(i) = ΔU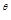
(ii) > ΔU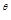
(iii) < ΔU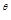
(iv) = 0
5. The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, -890.3 kJ 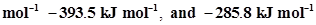 respectively. Enthalpy of formation of
respectively. Enthalpy of formation of 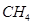 (g) will be
(g) will be
(i) –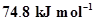
(ii) 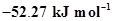
(iii)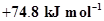
(iv)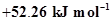 .
.
6. A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be
(i) possible at high temperature
(ii) possible only at low temperature
(iii) not possible at any temperature
(iv) possible at any temperature
7. In a process, 701 J of heat is absorbed by a system and 394 J ofwork is done by the system. What is the change in internal energy for the process?
8. The reaction of cyanamide,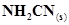 ,with dioxygen was carried out in a bomb calorimeter, and ΔU was found to be -742.7 kJ
,with dioxygen was carried out in a bomb calorimeter, and ΔU was found to be -742.7 kJ 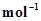 at 298 K. Calculate enthalpy change for the reaction at 298 K.
at 298 K. Calculate enthalpy change for the reaction at 298 K.
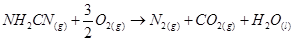
9. Calculate the number of kJ of heat necessary to raise the temperatureof 60.0 g of aluminium from 35°C to 55°C. Molar heat capacity of Al is 24 J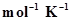 .
.
10. Calculate the enthalpy change on freezing of 1.0 mol of water at 10.0°C to ice at -10.0°C. 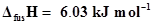 at 0°C.
at 0°C.
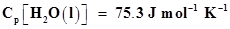
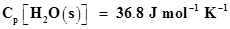
11. Enthalpy of combustion of carbon to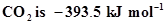 . Calculate the heat released upon formation of 35.2 g of
. Calculate the heat released upon formation of 35.2 g of 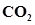 from carbon and dioxygen gas.
from carbon and dioxygen gas.
12. Enthalpies of formation of CO(g), 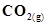 ,
, 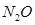 (g) and
(g) and 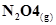 are
are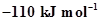 ,
, 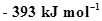 ,
, 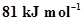 and 9.7 kJ
and 9.7 kJ 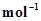 respectively. Find the value of Δ
respectively. Find the value of Δ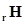 for the reaction:
for the reaction:
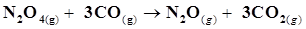
13. Given 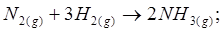 ;
; 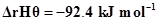
What is the standard enthalpy of formation of 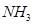 gas?
gas?
14. Calculate the standard enthalpy of formation of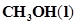 ) from the following data:
) from the following data:
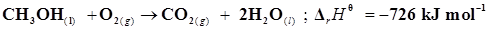
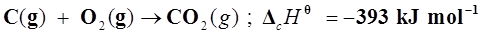
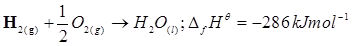
15. Calculate the enthalpy change for the process 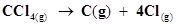 and calculate bond enthalpy of C-Cl in
and calculate bond enthalpy of C-Cl in .
.
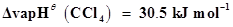 .
.
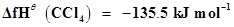 .
.
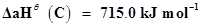 , where
, where 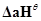 is enthalpy of atomisation
is enthalpy of atomisation
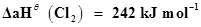
16. For an isolated system, ΔU = 0, what will be ΔS?
17. For the reaction at 298 K,
2A + B → C
ΔH = 400 kJ 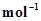 and ΔS = 0.2
and ΔS = 0.2 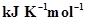
At what temperature will the reaction become spontaneous considering ΔH and ΔS to be constant over the temperature range?
18. For the reaction,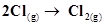 ,what are the signs of ΔH and ΔS ?
,what are the signs of ΔH and ΔS ?
19. For the reaction 2A(g) + B(g) → 2D(g) . Calculate 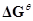 for the reaction, and predict whether the reaction may occur spontaneously.
for the reaction, and predict whether the reaction may occur spontaneously.
20. The equilibrium constant for a reaction is 10. What will be the value of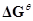 ? R = 8.314
? R = 8.314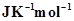 , T= 300 K.
, T= 300 K.
21. Comment on the thermodynamic stability of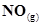 , given
, given
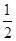
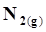 +
+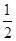
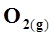
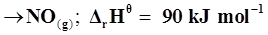
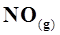 +
+ 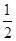
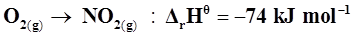
22. Calculate the entropy change in surroundings when 1.00 mol of 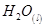 is formed under standard conditions.
is formed under standard conditions.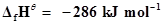 .
.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now