CBSE Question Paper 2017 class 12 Chemistry conducted by Central Board of Secondary Education, New Delhi in the month of March 2017. CBSE previous year question papers with the solution are available in the myCBSEguide mobile app and website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
CBSE Question Paper 2017 class 12 Chemistry

Class 12 Chemistry list of chapters
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d and f Block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday life
CBSE Question Paper 2017 class 12 Chemistry
General Instructions:
- All questions are compulsory.
- Question number 1 to 5 are very short-answer questions and carry 1 mark each.
- Question number 6 to 10 are short-answer questions and carry 2 marks each.
- Question number 11 to 22 are also short-answer questions and carry 3 marks each.
- Question number 23 is a value based question and carry 4 marks.
- Question number 24 to 26 are long-answer questions and carry 5 marks each.
- Use log tables, if necessary. Use of calculators is not allowed.
- What type of stoichiometric defect is shown by ZnS?
- What are emulsions? Give one example.
- Write IUPAC name of the complex: [CoCl2(en)2]+.
- What happens when phenol is oxidized by Na2Cr2O7/H2SO4?
- Write IUPAC name of the following compound:
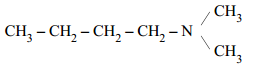
- Following reactions can occur at cathode during the electrolysis of aqueous silver nitrate solution using Pt electrodes:
Ag + (aq) + e – →Ag(s);Eo = 0.80VAg(aq) + + e – →Ag(s);Eo = 0.80V
H+aq+e−→12H2(g);E∘=0.00VHaq++e−→12H2(g);E∘=0.00V
On the basis of their standard electrode potential values, which reaction is feasible at cathode and why? - “Orthophosphoric acid (H3PO4) is not a reducing agent whereas hypophosphorous acid (H3PO2) is a strong reducing agent.” Explain and justify the above statement with the help of a suitable example.
- Explain why H2 and O2 do not react at room temperature.
- Write the rate equation for the reaction A2 + 3B2 →→ 2C, if the overall order of the reaction is zero.
OR
Derive integrated rate equation for rate constant of a first order reaction.
- Explain the following observations:
- Copper atom has completely filled d orbitals (3d10) in its ground state, yet it is regarded as a transition element.
- Cr2+ is a stronger reducing agent than Fe2+ in aqueous solutions.
- How will you carry out the following conversions:
- 2-Bromopropane to 1-bromopropane
- Benzene to p-chloronitrobenzene
- An element exists in bcc lattice with a cell edge of 288 pm. Calculate its molar mass if its density is 7.2 g/cm3.
- Calculate ΔrG∘ΔrG∘and log Kc for the following reaction at 298 K.
2Cr(s) + 3 Cd2+(aq)(aq)2+→ 2 Cr3+(aq)(aq)3+ + 3 Cd(s)
[Given: E°cell = + 0.34 V, IF = 96500C mol–1] - For a first order reaction, show that time required for 99% completion is twice the time required for completion of 90% reaction.
- Explain the following phenomenon giving reasons:
- Tyndall effect
- Brownian movement
- Physical adsorption decreases with increase in temperature.
- Write the principle involved in the following:
- Zone refining of metals
- Electrolytic refining
- Name the metal refined by each of the following processes:
- Mond Process
- van Arkel Method
- A mixed oxide of iron and chromium is fused with sodium carbonate in free access of air to form a yellow coloured compound (A). On acidification the compound (A) forms an orange coloured compound (B), which is a strong oxidizing agent. Identify compound (A) and (B). Write chemical reactions involved.
OR
- Give reasons for the following:
- Compounds of transition elements are generally coloured.
- MnO is basic while Mn2O7 is acidic.
- Calculate the magnetic moment of a divalent ion in aqueous medium if its atomic number is 26.
- Give reasons for the following:
- For the complex ion [Fe(en)2Cl2]+ write the hybridization type and magnetic behaviour. Draw one of the geometrical isomer of the complex ion which is optically active. [Atomic No.: Fe = 26]
- Account for the following:
- Electrophilic substitution reactions in haloarenes occur slowly.
- Haloalkanes, though polar, are insoluble in water.
- Arrange the following compounds in increasing order of reactivity towards SN2 displacement:
2–Bromo–2–Methylbutane, 1–Bromopentane, 2–Bromopentane
- Why phenol is more acidic than ethanol?
- Write the mechanism of acid dehydration of ethanol to yield ether:
2CH3CH2OH−→−−413kH+→413kH+CH3CH2OCH2CH3
- Account for the following:
- Identify A, B and C in the following reactions:
- CH3CH2Cl −→−−KCNA−→−−H2/NiB−→−−−−−−−−CH3COCI/BaseC→KCNA→H2/NiB→CH3COCI/BaseC
- C6H5N+22+2Cl– −→−−HBF4A−→−−−−−ΔNaNO2/CuB−→−−−Sn/HCIc→HBF4A→ΔNaNO2/CuB→Sn/HCIc
- Why water soluble vitamins must be supplied regularly in the diet? Give one example of it.
- Differentiate between the following:
- Essential and non-essential amino acids.
- Fibrous and globular proteins.
- Name a substance which can be used as an antiseptic as well as disinfectant.
- Name an artificial sweetener whose use is limited to cold foods and drinks.
- What are cationic detergents?
- Once there was a heavy downpour for about 3 hours in the early morning. Irfan and his family were finding it difficult to carry out their daily morning chores as the sewer water was flowing back into the toilets, the road in front of their house was flooded with water and they could not move out. On this very serious problem Irfan called a meeting of all the residents. In the meeting Irfan discussed the problem and said that we are using too much polythene bags and other plastic items which we throw here and there. All these move into the drains and sewer lines which get choked and do not allow flow of water. As these are non-biodegradable, they remain as such for a long time. So to overcome this problem, we should use bags made up of cloth or jute which are biodegradable.
Answer the following questions:- Name the polymer which is biodegradable. Write the structures of monomers and the repeating unit.
- Write two uses of this polymer.
- Write any two values shown by Irfan.
- Explain why on addition of 1 mol glucose to 1 litre water the boiling point of water increases.
- Henry’s law constant for CO2 in water is 1.67××108 Pa at 298 K. Calculate the number of moles of CO2 in 500 ml of soda water when packed under 2.53 ××105 Pa at the
-
OR
- Define the following terms:
- Ideal solution
- Osmotic pressure.
- Calculate the boiling point elevation for a solution prepared by adding 10 g CaCl2 to 200 g of water, assuming that CaCl2 is completely dissociated.same temperature.
(Kb for water = 0.512 K kgmol–1; Molar mass of CaCl2 = 111 g mol–1)
- When concentrated sulphuric acid was added to an unknown salt present in a test tube a brown gas (A) was evolved. This gas intensified when copper turnings were added to this test tube. On cooling the gas (A) changed into a colourless solid (B). Identify (A) and (B). Write chemical reactions involved.
- Draw structure of XeOF4.
- Define the following terms:
-
OR
- Account for the following:
- Reducing character decreases from SO2 to TeO2.
- HClO3 is a stronger acid than HClO.
- Xenon forms compounds with fluorine and oxygen only.
- Complete the following equations:
- 4NaCl + MnO2 + 4H2SO4 ––––
- 6XeF4 + 12H2O ––––
- Account for the following:
- Propanal is more reactive than propanone towards nucleophilic reagents.
- Electrophilic substitution in benzoic acid takes place at meta position.
- Carboxylic acids do not give characteristic reactions of carbonyl group.
- Give simple chemical test to distinguish between the following pairs of compounds:
- Acetophenone and benzaldehyde
- Benzoic acid and ethylbenzoate
- Account for the following:
-
OR
- Write structures of A, B, C and D in the following reaction sequence:
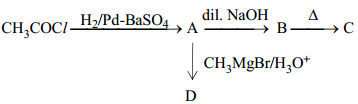
- Arrange the following compounds in the increasing order of their boiling points:
CH3CHO, CH3CH2OH, CH3OCH3, CH3COOH
- Write structures of A, B, C and D in the following reaction sequence:
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
Chemistry Question Paper 2017
Download class 12 Chemistry question paper with solution from best CBSE App the myCBSEguide. CBSE class 12 Chemistry question paper 2017 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper. CBSE question papers 2017 for class 12 Chemistry have 26 questions with solution.
Previous Year Question Paper for class 12 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 209, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Business Studies
- Economics
- History
- Geography
- Political Science
- Physical Education
- Computer Science
- Informatics Practices
- English Core
- Hindi Core
- Hindi Elective
- Other Subjects
To download CBSE Question Paper 2017 class 12 Accountancy, Chemistry, Physics, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now