NCERT Solutions class 12 Chemistry Chemical kinetics In text question Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for Chemical kinetics In text question as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER FOUR CHEMICAL KINETICS
- 4.1 Rate of a Chemical Reaction
- 4.2 Factors Influencing Rate of a Reaction
- 4.3 Integrated Rate Equations
- 4.4 Pseudo First Order Reaction
- 4.5 Temperature Dependence of the rate of a Reaction
- 4.6 Collision Theory of Chemical Reactions
NCERT Solutions class 12 Chemistry Chemical kinetics In text question In-text question
1. For the reaction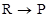 , the concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes. Calculate the average rate of reaction using units of time both in minutes and seconds.
, the concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes. Calculate the average rate of reaction using units of time both in minutes and seconds.
2. In a reaction, 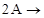 Products, the concentration of A decreases from
Products, the concentration of A decreases from 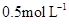 to
to 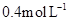 in 10 minutes. Calculate the rate during this interval?
in 10 minutes. Calculate the rate during this interval?
3. For a reaction,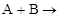 ’Product; the rate law is given by,
’Product; the rate law is given by, 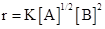 . What is the order of the reaction?
. What is the order of the reaction?
4. The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
5. A first order reaction has a rate constant 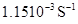 . How long will 5 g of this reactant take to reduce to 3 g?
. How long will 5 g of this reactant take to reduce to 3 g?
6. Time required to decompose 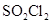 to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
to half of its initial amount is 60 minutes. If the decomposition is a first order reaction, calculate the rate constant of the reaction.
7. What will be the effect of temperature on rate constant?
8. The rate of the chemical reaction doubles for an increase of 10 K in absolute temperature from 298 K. Calculate 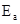 .
.
9. The activation energy for the reaction 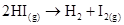 is
is 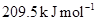 at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
at 581 K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide NCERT Solutions class 12 Chemistry Amines part 1
CBSE app for Class 12
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
NCERT Solutions for Class 12 Chemistry
Class 12 Chemistry NCERT book Solutions covers both organic and inorganic chemistry. Please note that CBSE has reduced the syllabus for class 12 Chemistry. So, you must check the latest syllabus of CBSE while going through the NCERT solution of any chapter given in the class 12 NCERT book.
- The Solid State part-1
- The Solid State part-2
- Solutions In-text question
- Solutions Part 1
- Solutions Part 2
- Solutions In-text question
- Electrochemistry In-text question
- Electrochemistry part 1
- Electrochemistry part 2
- Chemical kinetics In-text question
- Chemical kinetics part 1
- Chemical kinetics part 2
- Surface Chemistry In-text Questions
- Surface Chemistry part 1
- Surface Chemistry part 2
- General principle and processes of isolation of elements In-text Questions
- General principle and processes of isolation of elements part-1
- General principle and processes of isolation of elements part-2
- The p block elements part 1
- The p block elements part 2
- The p block elements In-text Questions
- The d and f block elements In-text Questions
- The d and f block elements part 1
- The d and f block elements part 2
- Coordination Compounds In-text Questions
- Coordination Compounds part 1
- Coordination Compounds part 2
- Haloalkanes and haloarenes In-text Questions
- Haloalkanes and haloarenes part 1
- Haloalkanes and haloarenes part 2
- Alcohol phenol and ether In-text Questions
- Alcohol phenol and ether part 1
- Alcohol phenol and ether part 2
- Aldehydes ketones and carboxylic acid In-text Questions
- Aldehydes ketones and carboxylic acid part 1
- Aldehydes ketones and carboxylic acid part 2
- Amines In-text Questions
- Amines part 1
- Amines part 2
- Biomolecules In-text Questions
- Biomolecules part 1
- Biomolecules part 2
- Polymers In-text Questions
- Polymers part 1
- Polymers part 2
- Chemistry in everyday life In-text Questions
- Chemistry in everyday life part 1
- Chemistry in everyday life part 2

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now