NCERT Solutions for Class 11 Chemistry Classification of Elements and Periodicity in Properties Part 1 Class 11 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 11 Chemistry chapter wise NCERT solution for Class 11 Chemistry Some Basic Concepts of Chemistry Part 1 and all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Class 11 Chemistry Download as PDF
NCERT Class 11 Chemistry Chapter wise Solutions
- Chapter 1 – Some Basic Concepts of Chemistry
- Chapter 2 – Structure of Atom
- Chapter 3 – Classification of Elements and Periodicity in Properties
- Chapter 4 – Chemical Bonding and Molecular Structure
- Chapter 5 – States of Matter
- Chapter 6 – Thermodynamics
- Chapter 7 – Equilibrium
- Chapter 8 – Redox Reactions
- Chapter 9 – Hydrogen
- Chapter 10 – The s-Block Elements
- Chapter 11 – The p-Block Elements
- Chapter 12 – Organic Chemistry – Some Basic Principles and Techniques
- Chapter 13 – Hydrocarbons
- Chapter 14 – Environmental Chemistry
NCERT Solutions for Class 11 Chemistry Classification of Elements and Periodicity in Properties Part 1
2. Which important property did Mendeleev use to classify the elements in his periodic table and did he stick to that?
3. What is the basic difference in approach between the Mendeleev’s Periodic Law and the Modern Periodic Law?
4. On the basis of quantum numbers, justify that the sixth period of the periodic table should have 32 elements.
5. In terms of period and group where would you locate the element with Z =114?
6. Write the atomic number of the element present in the third period and seventeenth group of the periodic table.
7. Which element do you think would have been named by
(i) Lawrence Berkeley Laboratory
(ii) Seaborg’s group?
8. Why do elements in the same group have similar physical and chemical properties?
9. What does atomic radius and ionic radius really mean to you?
10. How does atomic radius vary in a period and in a group? How do you explain the variation?
11. What do you understand by isoelectronic species? Name a species that will be
isoelectronic with each of the following atoms or ions.
(i) 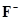
(ii) Ar
(iii) 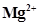
(iv) Rb+
12. Consider the following species:
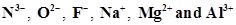
(a) What is common in them?
(b) Arrange them in the order of increasing ionic radii.
13. Explain why cations are smaller and anions larger in radii than their parent atoms?
14. What is the significance of the terms – ‘isolated gaseous atom’ and ‘ground state’ while defining the ionization enthalpy and electron gain enthalpy?
Hint: Requirements for comparison purposes.
15. Energy of an electron in the ground state of the hydrogen atom is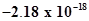 J. Calculate the ionization enthalpy of atomic hydrogen in terms of J mol-1.
J. Calculate the ionization enthalpy of atomic hydrogen in terms of J mol-1.
16. Among the second period elements the actual ionization enthalpies are in the order Li < B < Be < C < O < N < F < Ne.
Explain why
(i) Be has higher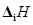 than B
than B
(ii) O has lower 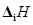 than N and F?
than N and F?
17. How would you explain the fact that the first ionization enthalpy of sodium is lower than that of magnesium but its second ionization enthalpy is higher than that of magnesium?
18. What are the various factors due to which the ionization enthalpy of the main group elements tends to decrease down a group?
19. The first ionization enthalpy values (in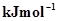 ) of group 13 elements are:
) of group 13 elements are:

How would you explain this deviation from the general trend?
20. Which of the following pairs of elements would have a more negative electron gain enthalpy?
(i) O or F (ii) F or Cl
NCERT Solutions for Class 11 Chemistry Chapter 3 Classification of Elements and Periodicity in Properties Part 1
NCERT Solutions Class 11 Chemistry Classification of Elements and Periodicity in Properties Part 1 PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 11 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 11 Chemistry have total 14 chapters. 11 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 11 solutions PDF and Chemistry ncert class 11 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
NCERT Solutions for Science Class 3rd to 12th
- NCERT Solutions for Class 12 Physics.
- NCERT Solutions for Class 12 Chemistry
- NCERT Solutions for Class 12 Biology
- NCERT solutions for class 11 Physics
- NCERT Solutions for Class 11 Chemistry
- NCERT solutions for class 11 Biology
- NCERT Solutions for Class 10 Science
- NCERT Solutions for Class 9 Science
- NCERT solutions for Class 8 Science
- NCERT solutions for Class 7 Science
- NCERT Solutions for Class 6 Science
- NCERT Solutions for Class 5 EVS
- NCERT Solutions for Class 4 EVS
- NCERT Solutions for Class 3 EVS
To download NCERT Solutions for class 11 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide.com website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now