NCERT Solutions for Class 11 Chemistry Some Basic Concepts of Chemistry Part 1 Class 11 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 11 Chemistry chapter wise NCERT solution for Class 11 Chemistry Some Basic Concepts of Chemistry Part 1 and all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Class 11 Chemistry Download as PDF
NCERT Class 11 Chemistry Chapter wise Solutions
- Chapter 1 – Some Basic Concepts of Chemistry
- Chapter 2 – Structure of Atom
- Chapter 3 – Classification of Elements and Periodicity in Properties
- Chapter 4 – Chemical Bonding and Molecular Structure
- Chapter 5 – States of Matter
- Chapter 6 – Thermodynamics
- Chapter 7 – Equilibrium
- Chapter 8 – Redox Reactions
- Chapter 9 – Hydrogen
- Chapter 10 – The s-Block Elements
- Chapter 11 – The p-Block Elements
- Chapter 12 – Organic Chemistry – Some Basic Principles and Techniques
- Chapter 13 – Hydrocarbons
- Chapter 14 – Environmental Chemistry
NCERT Solutions for Class 11 Chemistry Some Basic Concepts of Chemistry Part 1
(i) 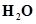
(ii) 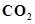
(iii) 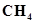
2. Calculate the mass percent of different elements present in sodium sulphate (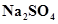 ).
).
3. Determine the empirical formula of an oxide of iron which has 69.9% iron and 30.1% dioxygen by mass.
4. Calculate the amount of carbon dioxide that could be produced when
(i) 1 mole of carbon is burnt in air.
(ii) 1 mole of carbon is burnt in 16 g of dioxygen.
(iii) 2 moles of carbon are burnt in 16 g of dioxygen.
5. Calculate the mass of sodium acetate 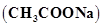 required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g
required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g 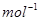
6. Calculate the concentration of nitric acid in moles per litre in a sample which has a density, 1.41 g 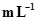 and the mass per cent of nitric acid in it being 69%.
and the mass per cent of nitric acid in it being 69%.
7. How much copper can be obtained from 100 g of copper sulphate (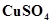 )?
)?
8. Determine the molecular formula of an oxide of iron in which the mass per cent of iron and oxygen are 69.9 and 30.1 respectively. Given that the molar mass of the oxide is 159.69 g 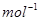 .
.
9. Calculate the atomic mass (average) of chlorine using the following data:

10. In three moles of ethane 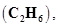 calculate the following:
calculate the following:
(i) Number of moles of carbon atoms.
(ii) Number of moles of hydrogen atoms.
(iii) Number of molecules of ethane.
11. What is the concentration of sugar (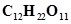 ) in mol
) in mol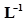 if its 20 g are dissolved in enough water to make a final volume up to 2 L?
if its 20 g are dissolved in enough water to make a final volume up to 2 L?
12. If the density of methanol is 0.793 kg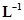 , what is its volume needed for making 2.5 L of its 0.25 M solution?
, what is its volume needed for making 2.5 L of its 0.25 M solution?
13. Pressure is determined as force per unit area of the surface. The SI unit of pressure, Pascal is as shown below: 1Pa = 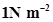 If mass of air at sea level is 1034 g
If mass of air at sea level is 1034 g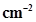 , calculate the pressure in Pascal.
, calculate the pressure in Pascal.
14. What is the SI unit of mass? How is it defined?
15. Match the following prefixes with their multiples:
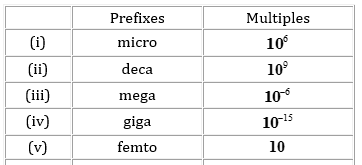
16. What do you mean by significant figures?
17. A sample of drinking water was found to be severely contaminated with chloroform, 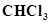 , supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
, supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
(i) Express this in percent by mass.
(ii) Determine the molality of chloroform in the water sample.
18. Express the following in the scientific notation:
(i) 0.0048
(ii) 234,000
(iii) 8008
(iv) 500.0
(v) 6.0012
19. How many significant figures are present in the following?
(i) 0.0025
(ii) 208
(iii) 5005
(iv) 126,000
(v) 500.0
(vi) 2.0034
NCERT Solutions for Class 11 Chemistry Chapter 1 Some Basic Concepts of Chemistry
NCERT Solutions Class 11 Chemistry Some Basic Concepts of Chemistry Part 1 PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 11 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 11 Chemistry have total 14 chapters. 11 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 11 solutions PDF and Chemistry ncert class 11 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
NCERT Solutions for Science Class 3rd to 12th
- NCERT Solutions for Class 12 Physics.
- NCERT Solutions for Class 12 Chemistry
- NCERT Solutions for Class 12 Biology
- NCERT solutions for class 11 Physics
- NCERT Solutions for Class 11 Chemistry
- NCERT solutions for class 11 Biology
- NCERT Solutions for Class 10 Science
- NCERT Solutions for Class 9 Science
- NCERT solutions for Class 8 Science
- NCERT solutions for Class 7 Science
- NCERT Solutions for Class 6 Science
- NCERT Solutions for Class 5 EVS
- NCERT Solutions for Class 4 EVS
- NCERT Solutions for Class 3 EVS
To download NCERT Solutions for class 11 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide.com website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now