NCERT Solutions for Class 11 Chemistry Chemical Bonding and Molecular Structure Part 2 Class 11 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 11 Chemistry chapter wise NCERT solution for Class 11 Chemistry Some Basic Concepts of Chemistry Part 1 and all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Class 11 Chemistry Download as PDF
NCERT Class 11 Chemistry Chapter wise Solutions
- Chapter 1 – Some Basic Concepts of Chemistry
- Chapter 2 – Structure of Atom
- Chapter 3 – Classification of Elements and Periodicity in Properties
- Chapter 4 – Chemical Bonding and Molecular Structure
- Chapter 5 – States of Matter
- Chapter 6 – Thermodynamics
- Chapter 7 – Equilibrium
- Chapter 8 – Redox Reactions
- Chapter 9 – Hydrogen
- Chapter 10 – The s-Block Elements
- Chapter 11 – The p-Block Elements
- Chapter 12 – Organic Chemistry – Some Basic Principles and Techniques
- Chapter 13 – Hydrocarbons
- Chapter 14 – Environmental Chemistry
NCERT Solutions for Class 11 Chemistry Chemical Bonding and Molecular Structure Part 2
Class 11 Chemistry Chemical Bonding and Molecular Structure Part 2
21. Apart from tetrahedral geometry, another possible geometry for 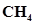 is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why
is square planar with the four H atoms at the corners of the square and the C atom at its centre. Explain why 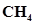 is not square planar?
is not square planar?
22. Explain why 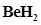 molecule has a zero dipole moment although the Be-H bonds are polar.
molecule has a zero dipole moment although the Be-H bonds are polar.
23. Which out of 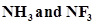 has higher dipole moment and why?
has higher dipole moment and why?
24. What is meant by hybridisation of atomic orbitals? Describe the shapes of sp, 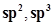 hybrid orbitals.
hybrid orbitals.
25. Describe the change in hybridisation (if any) of the Al atom in the followingreaction.
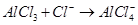
26. Is there any change in the hybridisation of B and N atoms as a result of the following reaction?
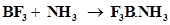
27. Draw diagrams showing the formation of a double bond and a triple bond between carbon atoms in 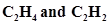 molecules.
molecules.
28. What is the total number of sigma and pi bonds in the following molecules?
(a) 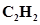
(b) 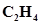
29. Considering x-axis as the internuclear axis which out of the following will notform a sigma bond and why? (a) 1s and 1s (b) 1s and 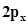 (c)
(c) 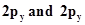 (d) 1s and 2s.
(d) 1s and 2s.
30. Which hybrid orbitals are used by carbon atoms in the following molecules?
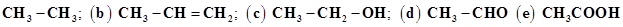
31. What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
32. Distinguish between a sigma and a pi bond.
33. Explain the formation of 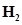 molecule on the basis of valence bond theory.
molecule on the basis of valence bond theory.
34. Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals.
35. Use molecular orbital theory to explain why the 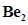 molecule does not exist.
molecule does not exist.
36. Compare the relative stability of the following species and indicate their magnetic properties;
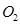 ,
,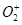 ,
,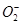 (superoxide),
(superoxide), 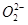 (peroxide)
(peroxide)
37. Write the significance of a plus and a minus sign shown in representing the orbitals.
38. Describe the hybridisation in case of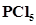 . Why are the axial bonds longer as compared to equatorial bonds?
. Why are the axial bonds longer as compared to equatorial bonds?
39. Define hydrogen bond. Is it weaker or stronger than the van der Waals forces?
40. What is meant by the term bond order? Calculate the bond order of: 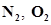 ,
, 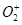 and
and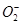 .
.
NCERT Solutions for Class 11 Chemistry Chapter 4 Chemical Bonding and Molecular Structure Part 2
NCERT Solutions Class 11 Chemistry Chemical Bonding and Molecular Structure Part 2 PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 11 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 11 Chemistry have total 14 chapters. 11 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 11 solutions PDF and Chemistry ncert class 11 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
NCERT Solutions for Science Class 3rd to 12th
- NCERT Solutions for Class 12 Physics.
- NCERT Solutions for Class 12 Chemistry
- NCERT Solutions for Class 12 Biology
- NCERT solutions for class 11 Physics
- NCERT Solutions for Class 11 Chemistry
- NCERT solutions for class 11 Biology
- NCERT Solutions for Class 10 Science
- NCERT Solutions for Class 9 Science
- NCERT solutions for Class 8 Science
- NCERT solutions for Class 7 Science
- NCERT Solutions for Class 6 Science
- NCERT Solutions for Class 5 EVS
- NCERT Solutions for Class 4 EVS
- NCERT Solutions for Class 3 EVS
To download NCERT Solutions for class 11 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide.com website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now