21.Discuss the chemistry of Lassaigne’s test.
22. Differentiate between the principle of estimation of nitrogen in an organic compound by (i) Dumas method and (ii) Kjeldahl’s method.
23. Discuss the principle of estimation of halogens, sulphur and phosphorus present in an organic compound.
24. Explain the principle of paper chromatography.
25. Why is nitric acid added to sodium extract before adding silver nitrate for testing halogens?
26. Explain the reason for the fusion of an organic compound with metallic sodium for testing nitrogen, sulphur and halogens.
27. Name a suitable technique of separation of the components from a mixture of calcium sulphate and camphor.
28.Explain, why an organic liquid vaporises at a temperature below its boiling point in its steam distillation?
29. Will 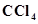 give white precipitate of AgCl on heating it with silver nitrate? Give reason for your answer.
give white precipitate of AgCl on heating it with silver nitrate? Give reason for your answer.
30. Why is a solution of potassium hydroxide used to absorb carbon dioxide evolved during the estimation of carbon present in an organic compound?
31. Why is it necessary to use acetic acid and not sulphuric acid for acidification of sodium extract for testing sulphur by lead acetate test?
32. An organic compound contains 69% carbon and 4.8% hydrogen, the remainder being oxygen. Calculate the masses of carbon dioxide and water produced when 0.20 g of this substance is subjected to complete combustion.
33. A sample of 0.50 g of an organic compound was treated according to Kjeldahl’s method. The ammonia evolved was absorbed in 50 mL of 0.5 M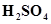 . The residual acid required 60 mL of 0.5 M solution of NaOH for neutralisation. Find the percentage composition of nitrogen in the compound.
. The residual acid required 60 mL of 0.5 M solution of NaOH for neutralisation. Find the percentage composition of nitrogen in the compound.
34.0.3780 g of an organic chloro compound gave 0.5740 g of silver chloride in Carius estimation. Calculate the percentage of chlorine present in the compound.
35. In the estimation of sulphur by Carius method, 0.468 g of an organic sulphur compound afforded 0.668 g of barium sulphate. Find out the percentage of sulphur in the given compound.
36. In the organic compound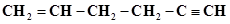 , the pair of hydridised orbitals involved in the formation of:
, the pair of hydridised orbitals involved in the formation of: 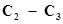 bond is:
bond is:
(a) 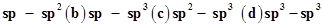
37. In the Lassaigne’s test for nitrogen in an organic compound, the Prussian blue colour is obtained due to the formation of:
(a) 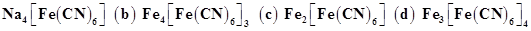
38.Which of the following carbocation is most stable?
(a) 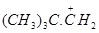
(b) 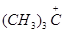
(c) 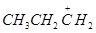
(d) 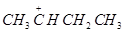
39. The best and latest technique for isolation, purification and separation of organic compounds is:
(a) Crystallisation
(b) Distillation
(c) Sublimation
(d) Chromatography
40. The reaction:
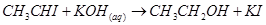
is classified as:
(a) electrophilic substitution
(b) nucleophilic substitution
(c) elimination
(d) addition

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now