Download CBSE Sample paper for class 09 Science from myCBSEguide. Sample paper for class 09 Science are available for download in myCBSEguide app, the best app for CBSE students. Sample Paper for class 09 Science includes questions from Science-NCERT Publication, Assessment of Practical Skills in Science-CBSE Publication, Laboratory Manual- Science-NCERT Publication, Exemplar Problems-NCERT Publication.
Download Complete set of sample paper for class 09 Science
For study on the go download myCBSEguide app for android phones. Sample paper for class 09 Science and other subjects are available for download as PDF in app too.
Science Sample papers
Here is Sample paper for class 09 Science. To get the answers and more sample papers, visit myCBSEguide App or website stated above.

Sample Paper for class 09 Science
Session 2017-2018
General Instructions:
- The question paper comprises of three Sections A, B and C. You are to attempt all the sections.
- All questions are compulsory.
- There is no choice in any of the questions.
- All questions of Section A, Section B are to be attempted separately.
- Question numbers 1 to 2 in Section A are one mark questions. These are to be answered in one word or in one sentence.
- Question numbers 3 and 5 in Section A are two marks questions. These are to be answered in about 30 words each.
- Question numbers 6 to 15 in Section A are three marks questions. These are to be answered in about 50 words each.
- Question numbers 16 to 21 in Section A are fve marks questions. These are to be answered in about 70 words each.
- Question numbers 22 to 27 in Section B are two marks questions based on practical skills. These are to be answers in about 30 words each.
Section ‘A’
- A person travels 6 Km. in a circular path and reaches back on the starting point. Calculate the displacement.
Ans. Displacement is zero because starting point of the journey and ending point of the journey is same. - Write names of solutions used to stain plant cell and animal cell white making a slide.
Ans. Safranine is used to stain plant cell.
Methylene blue is used to stain animal cell. - Recognise the phylum of animals on the basis of characteristics given. below. Also write one examples of each.Ans.
- Coelom is filled with blood and legs and jointed.
- These animal are free living or parasite and these are called flat worms.
- Phylum-Arthropoda-eg-Housefly, butterfly, spider etc. The animals have coelom filled with blood and jointed legs.
- Platyhelminthes is the phylum of flat worms. They are free living or parasite. example: planaria, liver fluke.
- How does endocytosis help is single celled organism like amaeba?
Ans. Amoeba is a unicellular organism. All the processes of living being are performed in this organism. Plasma membrane is flexible. So amoeba can take its food from surroundings. This process is called endocytosis. - Write one word for the following:-
- A group of cells having structure in which cells are arrnaged in aspecial order to perform a work.
- The process of taking permanent shape and size to perform a special function.
- Animal tissue which attaches muscles to bone.
- Kindly shaped cells surrounding the stomata.
Ans.
(a) Tissue
(b) Differentiation
(c) Tendors
(d) Guard cells. - (a) Explain the law of conservation of energy.
(b) Give one example of each energy transformation:-
(i) Heat energy to mechanical energy.
(ii) Mechanical energy to electric energy.
(c) A person is standing from 20 minutes with heavy weight on his head. Does he do any work or not? explain.
Ans. (a) According to conservation of energy, “Energy can neither be created nor destroyed”. It can be transformed from one form to it’s another form. Total energy before and after transformation remains the same.
(b) (i) Heat is formed by burning of fuel in an engine. This heat energy is converted into mechanical energy.
(ii) In an electric generator. Mechanical energy is converted into electrical energy. - (a) An electric heater of 2000 Walt is working for 5 hours. How much energy it consume?
(b) Convert commercial unit of energy into S.I. unit of energy.
Ans. (a) Power of electric heater (P) = 2000 w
Time of use (T) = 5 Hr
Consumed energy (E) = P × t = 2000 × 5 wh = {tex}\frac{{2000}}{{1000}}{/tex} kw × 5h = 10 Kwh.
(b) Commercial unit of energy = 1 Kwh
S.I. unit of energy = 1 Joule
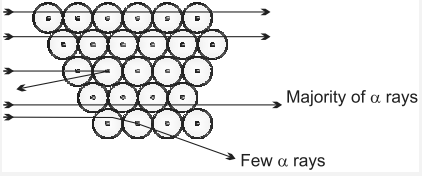
1 Kwh = 1000 × 60 × 60 ws = 3.6 × 106 Joules. - (a) Draw a diagram showing Rutherford’s {tex}\alpha {/tex} rays scattering experiments”.
(b) Why Rutherford used gold foil in above said experiment?
Ans. (a)
(b) Rutherford choose gold foil because they want a very thin layer. Thickness of gold foil is equal to the thickness of 1000 atom put together. - The number of neutrons and protons in the nuclei of two atoms A and B is as follow :
A B Proton 8 8 Nuetron 8 10 (a) Write atomic mass of A and B.
(b) Write the relationship between A and B.
(c) Write electronic configuration of atom A and B.Ans. (a) Both A and B have same number of electrons, so they have same electronic configuration = 8 = 2, 6
- Differentiate the following (Write one difference)
(a) Gynmosperm and angiosperm
(b) Birds (Aves) and mammlas.
(c) Monocotyletons and dicotyledons plants.
Ans. (a) Angiosperm are flowering plants in which seeds are found inside the fluits. In gymnosperms seeds are naked.
(b) Birds are oviparous animals with dry skin covered by feathers and wings. Their forelimbs are modified into wings. Mammals are viviparous (except some exceptions) with sweat and oil glands on their skin.
(c) In monocotyledons plants seeds contain only one cotyledon while in dicotyledons plants seeds contain two cotyledons. - What is formula unit mass? How does it calculated?
Calculate the formula unit mass of a compound Na2S2O3. (Na=23u, S=32u, O=16u)
Ans. Formula Unit Mass:- It is the sum of atomic mass of ions of atoms present in formula for a compound.
Formula unit mass of Na2S2O3 = (2 × Na atomic mass) + (2 × S atomic mass) + (3 × O atomic mass)
= (2 × 23) + (2 × 32) + (3 × 16)
= 1584 u. - Give Reasons:-
(a) When a ball dropped from a height, its speed continuously increased.
(b) When we stop pedaling a bicycle, its speed slows down.
(c) When a tree is shaken vigorously, its fruits and leaves fall down.
Ans. (a) When a ball is droped from a height. Its speed continuously increased due to force of gravity (acceleration due to gravity increase on coming downwards).
(b) When we stop pedaling a bicycle, its speed slows down due to air resistance and force of friction between type of bicycle and surface of road. So retardation force is created which stop the bicycle.
(c) When tree is shaken. It moves to and slightly but its fruits and leave tends to remain at rest due to their inertia and hence detached from the tree and fall down. - Cattle farming. poultry and fish farming are included in animal husbandry. What is the need of animal husbandry.
Ans. Management of cattle farming is known as animal husbandry. It includes various tasks such as giving food to cattles, animals, shelter, breeding, health, disease control and proper economic utilization. Animal farming includes cow, buffalo, goat, sheep, camel, poultry, and fisheries. Due to increasing population, demand for milk, eggs, meat etc is also increased. Now farmers take up animal husbandry on commercial basis so it needs scientific management practices. cattle farming is done for milk and agricultural work. By poultry we increase meat an eggs production. fisheries is a cheap source of protein food. - (a) Meristematic cell contains dense or abundant cytoplasm a single large nucleus but no vacuole.
(b) In sclerenchyma tissue intercellular space is absent in between the cells.
(c) In xerophytic plants, epidermis is covered with cutin.
Give Reason:-Ans. (a) Cells of meristems divide continuously and help in increasing the length and girth of the plants, so they have abundant cytoplasm and prominent nucleus, thin cell wall but no vacuoles. Vacuole is like a bag which store or liquid substances. Meristematic cells do not have stared food in them.
(b) In sclerenchyma tissue intercellular space is absent in between the cells because these cells are dead and cell walls are thicked due to deposition of lignin which acts as cement.
(c) In xerophyte plants, epidermis is covered with cutin. Cutin is a layer which resists transport of water through it. Desert have a little water. Cutin protects cells form dryness and also check transpiration from leaves. - A bike is moving with a velocity of 90 Km/hr. After applying break it stop in 5 sec. Calculate the force applied by breaks. Mass of motorcycle and rides is 200 Kg.
Ans. Mass of motorcycle and rider m = 200 Kg
Initial velocity u = 90 Km/h-1 {tex} = 90 \times \frac{5}{{18}}{/tex}
Final Velocity of motor cycle v = 0
Time (t) = 5s
Acceleration {tex}a = \frac{{u – v}}{t}{/tex}
Force F = ma
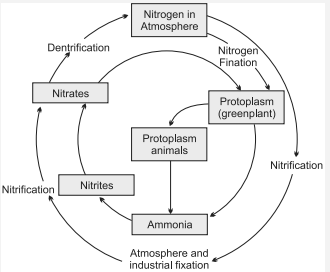
{tex}F = \frac{{200 \times (0 – 25)}}{5} = – 1000N{/tex} - With the help of a labelled diagram show nitrogen cycle in nature. Name two molecules essential to life which contain nitrogen. Explain any two processes involved in the cycling of nitrogen in the environment.
Ans. Various steps of nitrogen cycle are :
Two molecules essential to life which contain nitrogen are:
(i) Nucleic acids
(ii) Some vitamins.
Two processes involved in the cycling of nitrogen in the environment are:
1. Nitrogen fixing bacteria are formed in the roots of legumes. These bacteria fix atmospheric nitrogen in the form of nitrates and nitrites which can then be used by plants.
2. Fixation of nitrogen by lightning-During lightning, the high temperatures and pressures created in the air convert nitrogen into oxides of nitrogen. These oxides dissolve in water to give nituie and nitrogen acids and fall on land along with rain. These are then utilised by various like forms. - (a) Name three diseases against which immunisation vaccines are available.
(b) State in brief the principle of immunisation.
(c) Why do we not need to immune many infants in our country (below the age of 5) against Hepatitis A?
Ans. (a) 1. Polio.
2. Measles
3. Tetanus
(b) When the immune system first seen an infections microbe, it responds against this and then remembers it specifically. The next time when that particular microbe or its close relatives enter the body, the immune system responds more vigorously. This eliminates the infection more quickly than the first time around. This is the principle of immunisation.
(c) Because they have already been exposed to hepatitis. A virus through contaminated water and gone immune to it. - What is:
(a) Wave Length
(b) Frequency
(c) Amplitude.
Write its S.I. units. Derive the relationship between speed, wavelength and frequency.
Ans. (a) Wave Length: The distance between two consecutive compressions (C) or two consecutive rare factions (R) is called wavelength. The wave length is represented by {tex}\lambda{/tex} (Lambda). Its S.I. unit is meter (m).
(b) Frequency: The number of oscillations per unit time is called frequency of the sound wave. It is represented by v (nu). Its S.I. unit is hertz (Hz).
(c) Amplitude: The maximum displacement of the particles of the medium from their original undisturbed position, When a wave passes through a medium is called amplitude. It is represented by the letter A. The SI unit of amplitude of sound wave is the unit of density.
Relationship between speed, wavelength and frequency
Distance travelled m/s is speed
V = {tex}\frac{{{\text{Distance travelled by one complete wave}}}}{{{\text{Time taken to complete one wave}}}}{/tex}
{tex}{\text{ = }}\frac{\lambda }{{Ts}}{/tex}
{tex}V = \frac{\lambda }{T} = \lambda v\;\left( {\frac{1}{T} = v} \right){/tex}
V = λv
Wave speed = wave length × frequency - (a) What is the physical state of water at:
(i) 250ºC (ii) 100ºC (iii) 0ºC
(b) Convert the following temperature to the celsius selae:
(i) 298 k (ii) 300 k (iii) 280 k
Ans. (a)
(i) 250ºC – Gas
(ii) 100ºC – Gas
(iii) 0ºC – Solid
(b)
(i) 298 k = 298 – 273 = 25ºC
(ii) 300 k = 300 – 273 = 27ºC
(iii) 280 k = 280 – 273 = 7ºC - A student was given mixture of iron filling and sulphur on heating the mixture. Observe the following:
(a) What was the colour of compound.
(b) Write magnetic effect on the mixture.
(c) Write the reaction between carbon disulphide and mixture.
(d) On adding dilute sulphuric acid or dilute, hydrochloric acid what will happen?
Name the gas obtained and write two properties.
Ans. (a) When mixture of iron filling and sulphur is reacted iron sulphide is obtained which is black in colour.
(b) Iron does not get attracted to a magnet from a mixture of reacted iron fillings and sulphur powder.
(c) No reaction on carbon disulphide and mixture.
(d) On adding dilute hydrochloride acid. The gas obtained is hydrogen sulphide (H2S).
(i) It is colourless gas.
(ii) It smells like rotten eggs. - (i) If a man jumps out from a boat, the boat moves backwards. Why?
(ii) Explain our walking in terms of Newton’s third law of motion.
(iii)A bullet of mass 10 g travelling horizontally with a velocity of 150 ms-1 strikes a stationary wooden block and comes to rest in 0.03 S.
Calculate the distance of penetration of the bullet into the block.
Ans. (i) When a man jumps out of a boat to the bank of the river, he applies a force in the forward direction. Due to the reaction of this boat moves backward (i.e. away from the bank).
(ii) When we walk on the ground, our foot pushes the ground backward (action) and in return the ground pushes our foot forward (reaction).
The forward reaction enerted by the ground on our foot makes us walk forward.
(iii) Mass of bullet m = 10 g = {tex}\frac{{10}}{{1000}}{/tex} kg= 0.01 kg
Initial velocity, u = 150 ms-1
Final velocity, v = 0
Time taken = 0.03 sec
Then, acceleration.
{tex}a = \frac{{v – u}}{t} = \frac{{(0 – 150)}}{{0.03}} = – 500\,m{s^{ – 2}}{/tex}
By third law of motion, v2 – u2 = 2as
{tex}s = \frac{{{v^2} – {u^2}}}{{2a}}{/tex} {tex}= \frac{{{{(0)}^2} – {{(150)}^2}}}{{2 \times ( – 5000)}} = \frac{{ – 150 \times 150}}{{ – 2 \times 5000}}{/tex}
= 2.25 m
SECTION ‘B’
- Mention the type of thermometer that should be used to determine the melting point of ice in laboratory? What should be the position of bulb in thermometer?
Ans. Thermometer of range of 0-100 mL is used to determine the melting point of ice laboratory. Bulb of thermometer should remain the ice cubes. - Deepti was asked to prepare four separate mixtures in four beakers A, B, C, & D by mixing sugar, fne sand, thin paste of starch and chalk powder respectively in water and then categories each as stable or unstable. What will be the correct categorization?
Ans. Sugar (stable), fine sand (unstable), thin paste of starch (stable) and chalk powder (unstable). - Fore limbs of birds are modified. Give reasons.
Ans. Fore limbs of birds are modified into using for flying. They have an outer covering of feathers and hollow bones that help them to fly. - Name the technique by which we can prepare a colloidal solution of starch in water.
Ans. Take about 0.5 gm of starch in a china dish. Pour about 10 mL of distilled water. stir the contents with a glass tube continuously. Heat the water. In this way colloidal solution of starch in water is formed. - Mention the two precaution to determine the boiling point of water in laboratory.
Ans. (i) The position of bulb should be slightly above the water level.
(ii) Stir the water continuously in beaker so that temperature should remain constant. - Four students A, B, C, & D in a group take 6m long slinky and produce a longitudinal wave, they observe that in 10 sec, 4 waves completed. On these observation the speed of wave in slinky is
(a) 4.8 m/s (b) 6.4 m/s (c) 7.2 m/s (d) 9.6 m/s
Ans. (a) 4.8 m/s
Explanation:
Total distance covered by wave in slinky to cover 4 forth backward motion
S = 4 × 2 × 6 = 48 m
Total time taken (t) = 10 s
{tex}{\text{Speed = }}\frac{{{\text{Distance}}}}{{{\text{Time}}}}{/tex}
{tex} = \frac{{48m}}{{10s}}{/tex}
= 4.8 ms-1
This is only initial part of the whole sample paper. Download Complete set of sample paper for class 09 Science
Sample Paper for class 09
It is Sample paper for class 09 Science. However, myCBSEguide provides the best sample papers for all the subjects. There are number of sample papers which you can download from myCBSEguide website. Sample paper for class 09 all subjects
- CBSE Sample Papers for Class 9 Mathematics
- CBSE Sample Papers for Class 9 Science
- CBSE Sample Papers for Class 9 Social Science
- CBSE Sample Papers for Class 9 English Communicative
- CBSE Sample Papers for Class 9 English Language and Literature
- CBSE Sample Papers for Class 9 Hindi Course A
- CBSE Sample Papers for Class 9 Hindi Course B
These are also Sample Paper for class 09 Science available for download through myCBSEguide app. These are the latest Sample Paper for class 09 Science as per the new exam pattern. Download the app today to get the latest and up-to-date study material.
Marking Scheme for Class 09 exam
| Subject | Annual Exam | Internal Assessment | Total Marks |
| English | 80 Marks | 20 Marks | 100 Marks |
| Hindi | 80 Marks | 20 Marks | 100 Marks |
| Mathematics | 80 Marks | 20 Marks | 100 Marks |
| Science | 80 Marks | 20 Marks | 100 Marks |
| Social Science | 80 Marks | 20 Marks | 100 Marks |
| Sanskrit | 80 Marks | 20 Marks | 100 Marks |
| Foundation of IT | 40 Marks | 60 Marks | 100 Marks |
Download CBSE Sample Papers
To download complete sample paper for class 09 Science, Hindi A, Hindi B, English Language, Social Science, English Communicative and Maths; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now