
myCBSEguide App
Download the app to get CBSE Sample Papers 2025-26, NCERT Solutions (Revised), Most Important Questions, Previous Year Question Bank, Mock Tests, and Detailed Notes.
Install NowNCERT Solutions class 12 Chemistry The d and f block elements In text Questions solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for The d and f block elements In text Questions as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER EIGHT THE D-AND F-BLOCKS ELEMENTS
- 8.1 Position in the Periodic Tabble
- 8.2 Electronic Configurations of the d-Block Elements
- 8.3 General Properties of the Transition Elements (d-Block)
- 8.4 Some important Compounds of Transition Elements
- 8.5 The Lanthanoids
- 8.6 The Actinoids
- 8.7 Some Applications of d-and f-Block Elements
NCERT Solutions class 12 Chemistry The d and f block elements In text Questions
In-text question
1. Silver atom has completely filled d orbitals 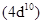 in its ground state. How can you say that it is a transition element?
in its ground state. How can you say that it is a transition element?
2. In the series Sc (Z = 21) to Zn (Z = 30), the enthalpy of atomization of zinc is the lowest, i.e., 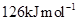 . Why?
. Why?
3. Which of the 3d series of the transition metals exhibits the largest number of oxidation states and why?
4. The 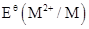 value for copper is positive (+0.34V). What is possibly the reason for this? (Hint: consider its high
value for copper is positive (+0.34V). What is possibly the reason for this? (Hint: consider its high 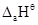 and low
and low 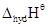 )
)
5. How would you account for the irregular variation of ionization enthalpies (first and second) in the first series of the transition elements?
6. Why is the highest oxidation state of a metal exhibited in its oxide or fluoride only?
7. Which is a stronger reducing agent 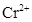 or
or 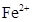 and why?
and why?
8. Calculate the ‘spin only’ magnetic moment of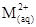 ion (Z = 27).
ion (Z = 27).
9. Explain why 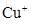 ion is not stable in aqueous solutions?
ion is not stable in aqueous solutions?
10. Actinoid contraction is greater from element to element than lanthanoid contraction. Why?
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now