NCERT Solutions class 12 Chemistry Solutions Part 1 Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for Solutions Part-1 as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER TWO SOLUTIONS
- 2.1 Types of Solutions
- 2.2 Expressing Concentration of Solutions
- 2.3 Solubility
- 2.4 Vapour Pressure of Liquid Solutions
- 2.5 Ideal and Non-Ideal Solutions
- 2.6 Colligative Properties and Determination of Molar Mass
- 2.7 Abnormal Molar Masses
NCERT Solutions class 12 Chemistry Solutions Part 1
1. Define the term solution. How many types of solutions are formed? Write briefly about each type with an example.
2. Give an example of solid solution in which the solute is a gas.
3. Define the following terms:
(i) Mole fraction
(ii) Molality
(iii) Molarity
(iv) Mass percentage.
4. Concentrated nitric acid used in laboratory work is 68% nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is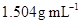 ?
?
Molarity of solution = 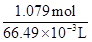 = 16.23 M
= 16.23 M
5. A solution of glucose in water is labelled as 10% w/w, what would be the molality and mole fraction of each component in the solution? If the density of solution is 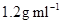 , then what shall be the molarity of the solution?
, then what shall be the molarity of the solution?
6. How many mL of 0.1 M HCl are required to react completely with 1 g mixture of 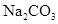 and
and 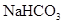 containing equimolar amounts of both?
containing equimolar amounts of both?
7. A solution is obtained by mixing 300g of 25% solution and 400 g of 40% solution by mass. Calculate the mass percentage of the resulting solution.
8. An antifreeze solution is prepared from 222.6 g of ethylene glycol (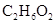 ) and 200 g of water. Calculate the molality of the solution. If the density of the solution is
) and 200 g of water. Calculate the molality of the solution. If the density of the solution is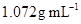 , then what shall be the molarity of the solution?
, then what shall be the molarity of the solution?
9. A sample of drinking water was found to be severely contaminated with chloroform (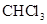 ) supposed to be a carcinogen. The level of contamination was 15 ppm (by mass):
) supposed to be a carcinogen. The level of contamination was 15 ppm (by mass):
(i) express this in percent by mass
(ii) determine the molality of chloroform in the water sample.
10. What role does the molecular interaction play in a solution of alcohol and water?
11. Why do gases always tend to be less soluble in liquids as the temperature is raised?
12. State Henry’s law and mention some important applications?
13. The partial pressure of ethane over a solution containing 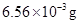 of ethane is 1 bar. If the solution contains
of ethane is 1 bar. If the solution contains 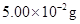 of ethane, then what shall be the partial pressure of the gas?
of ethane, then what shall be the partial pressure of the gas?
4. What is meant by positive and negative deviations from Raoult’s law and how is the sign of 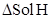 related to positive and negative deviations from Raoult’s law?
related to positive and negative deviations from Raoult’s law?
15. An aqueous solution of 2% non-volatile solute exerts a pressure of 1.004 bar at the normal boiling point of the solvent. What is the molar mass of the solute?
16. Heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components are 105.2 kPa and 46.8 kPa respectively. What will be the vapour pressure of a mixture of 26.0 g of heptane and 35 g of octane?
17. The vapour pressure of water is 12.3 kPa at 300 K. Calculate vapour pressure of 1 molal solution of a non-volatile solute in it.
18. Calculate the mass of a non-volatile solute (molar mass 40 g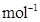 ) which should be dissolved in 114 g octane to reduce its vapour pressure to 80%.
) which should be dissolved in 114 g octane to reduce its vapour pressure to 80%.
19. A solution containing 30 g of non-volatile solute exactly in 90 g of water has a vapour pressure of 2.8 kPa at 298 K. Further, 18 g of water is then added to the solution and the new vapour pressure becomes 2.9 kPa at 298 K. Calculate:
i. molar mass of the solute
ii. vapour pressure of water at 298 K.
20. A 5% solution (by mass) of cane sugar in water has freezing point of 271 K. Calculate the freezing point of 5% glucose in water if freezing point of pure water is 273.15 K.
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now