NCERT Solutions class 12 Chemistry The d and f block elements part 2 Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for The d and f block elements part 2 as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER EIGHT THE D-AND F-BLOCKS ELEMENTS
- 8.1 Position in the Periodic Tabble
- 8.2 Electronic Configurations of the d-Block Elements
- 8.3 General Properties of the Transition Elements (d-Block)
- 8.4 Some important Compounds of Transition Elements
- 8.5 The Lanthanoids
- 8.6 The Actinoids
- 8.7 Some Applications of d-and f-Block Elements
NCERT Solutions class 12 Chemistry The d and f block elements part 2
21. How would you account for the following:
(i) Of the 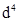 species,
species, 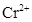 is strongly reducing while manganese(III) is strongly oxidising.
is strongly reducing while manganese(III) is strongly oxidising.
(ii) Cobalt(II) is stable in aqueous solution but in the presence of complexing reagents it is easily oxidised.
(iii) The 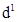 configuration is very unstable in ions.
configuration is very unstable in ions.
22. What is meant by ‘disproportionation’? Give two examples of disproportionation reaction in aqueous solution.
(i) 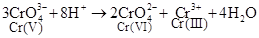
Cr(V) is oxidized to Cr(VI) and reduced to Cr(III).
(ii) 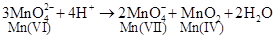
Mn (VI) is oxidized to Mn (VII) and reduced to Mn (IV).
23. Which metal in the first series of transition metals exhibits +1 oxidationstate most frequently and why?
24. Calculate the number of unpaired electrons in the following gaseous ions: 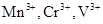 and
and 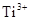 . Which one of these is the most stable in aqueous solution?
. Which one of these is the most stable in aqueous solution?
25. Give examples and suggest reasons for the following features of the transition metal chemistry:
(i) The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
(ii) A transition metal exhibits highest oxidation state in oxides and fluorides.
(iii) The highest oxidation state is exhibited in oxoanions of a metal.
26. Indicate the steps in the preparation of:
(i) 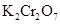 from chromite ore.
from chromite ore.
(ii) 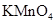 from pyrolusite ore.
from pyrolusite ore.
27. What are alloys? Name an important alloy which contains some of the lanthanoid metals. Mention its uses.
28. What are inner transition elements? Decide which of the following atomic numbers are the atomic numbers of the inner transition elements: 29, 59, 74, 95, 102, 104.
29. The chemistry of the actinoid elements is not so smooth as that of the Lanthanoids. Justify this statement by giving some examples from the oxidation state of these elements.
30. Which is the last element in the series of the actinoids? Write the electronic configuration of this element. Comment on the possible oxidation state of this element.
31. Use Hund’s rule to derive the electronic configuration of 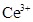 ion and calculate its magnetic moment on the basis of ‘spin-only’ formula.
ion and calculate its magnetic moment on the basis of ‘spin-only’ formula.
32. Name the members of the lanthanoid series which exhibit +4 oxidation state and those which exhibit +2 oxidation state. Try to correlate this type of behavior with the electronic configurations of these elements.
33. Compare the chemistry of the actinoids with that of lanthanoids with reference to:
(i) electronic configuration
(ii) oxidation states and
(iii) chemical reactivity.
34. Write the electronic configurations of the elements with the atomic numbers 61, 91, 101, and 109.
35. Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following points:
(i) electronic configurations,
(ii) oxidation states,
(iii) ionisation enthalpies, and
(iv) atomic sizes.
36. Write down the number of 3d electrons in each of the following ions: 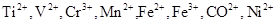 and
and 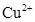 . Indicate how would you expect the five 3d orbitals to be occupied for these hydrated ions (octahedral).
. Indicate how would you expect the five 3d orbitals to be occupied for these hydrated ions (octahedral).
37. Comment on the statement that elements of the first transition series possess many properties different from those of heavier transition elements.
38. What can be inferred from the magnetic moment values of the following complex species?
Example Magnetic Moment (BM)
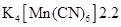
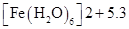
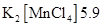
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
NCERT Solutions for Class 12 Chemistry
Class 12 Chemistry NCERT book Solutions covers both organic and inorganic chemistry. Please note that CBSE has reduced the syllabus for class 12 Chemistry. So, you must check the latest syllabus of CBSE while going through the NCERT solution of any chapter given in the class 12 NCERT book.
- The Solid State part-1
- The Solid State part-2
- Solutions In-text question
- Solutions Part 1
- Solutions Part 2
- Solutions In-text question
- Electrochemistry In-text question
- Electrochemistry part 1
- Electrochemistry part 2
- Chemical kinetics In-text question
- Chemical kinetics part 1
- Chemical kinetics part 2
- Surface Chemistry In-text Questions
- Surface Chemistry part 1
- Surface Chemistry part 2
- General principle and processes of isolation of elements In-text Questions
- General principle and processes of isolation of elements part-1
- General principle and processes of isolation of elements part-2
- The p block elements part 1
- The p block elements part 2
- The p block elements In-text Questions
- The d and f block elements In-text Questions
- The d and f block elements part 1
- The d and f block elements part 2
- Coordination Compounds In-text Questions
- Coordination Compounds part 1
- Coordination Compounds part 2
- Haloalkanes and haloarenes In-text Questions
- Haloalkanes and haloarenes part 1
- Haloalkanes and haloarenes part 2
- Alcohol phenol and ether In-text Questions
- Alcohol phenol and ether part 1
- Alcohol phenol and ether part 2
- Aldehydes ketones and carboxylic acid In-text Questions
- Aldehydes ketones and carboxylic acid part 1
- Aldehydes ketones and carboxylic acid part 2
- Amines In-text Questions
- Amines part 1
- Amines part 2
- Biomolecules In-text Questions
- Biomolecules part 1
- Biomolecules part 2
- Polymers In-text Questions
- Polymers part 1
- Polymers part 2
- Chemistry in everyday life In-text Questions
- Chemistry in everyday life part 1
- Chemistry in everyday life part 2

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now