1. What are the common physical and chemical features of alkali metals?
2. Discuss the general characteristics and gradation in properties of alkaline earth metals.
3. Why are alkali metals not found in nature?
4. Find the oxidation state of sodium in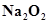
5. Explain why is sodium less reactive than potassium?
6. Compare the alkali metals and alkaline earth metals with respect to (i) ionization enthalpy (ii) basicity of oxides and (iii) solubility of hydroxides.
7. In what ways lithium shows similarities to magnesium in its chemical behaviour?
8. Explain why alkali and alkaline earth metals cannot be obtained by chemical reduction methods?
9. Why are potassium and cesium, rather than lithium used in photoelectric cells?
10. When an alkali metal dissolves in liquid ammonia the solution can acquire different colours. Explain the reasons for this type of colour change.
11. Beryllium and magnesium do not give colour to flame whereas other alkaline earth metals do so. Why?
12. Discuss the various reactions that occur in the Solvay process.
13. Potassium carbonate cannot be prepared by Solvay process. Why?
14. Why is 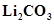 decomposed at a lower temperature whereas
decomposed at a lower temperature whereas 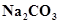 at higher temperature?
at higher temperature?
15. Compare the solubility and thermal stability of the following compounds of the alkali metals with those of the alkaline earth metals. (a) Nitrates (b) Carbonates (c) Sulphates.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now