
myCBSEguide App
Download the app to get CBSE Sample Papers 2024-25, NCERT Solutions (Revised), Most Important Questions, Previous Year Question Bank, Mock Tests, and Detailed Notes.
Install Now1. Discuss the pattern of variation in the oxidation states of
(i) B to Tl and
(ii) C to Pb.
2. How can you explain higher stability of 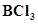 as compared to
as compared to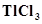 ?
?
3. Why does boron trifluoride behave as a Lewis acid?
4. Consider the compounds,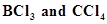 . How will they behave with water? Justify.
. How will they behave with water? Justify.
5. Is boric acid a protic acid? Explain.
6. Explain what happens when boric acid is heated.
7. Describe the shapes of 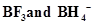 . Assign the hybridisation of boron in these species.
. Assign the hybridisation of boron in these species.
8. Write reactions to justify amphoteric nature of aluminium.
9. What are electron deficient compounds? Are 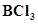 and
and 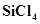 electron deficient species? Explain.
electron deficient species? Explain.
10. Write the resonance structures of 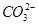 and
and 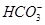 .
.
11. What is the state of hybridisation of carbon in (a) 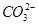 (b) diamond (c) graphite?
(b) diamond (c) graphite?
12. Explain the difference in properties of diamond and graphite on the basis of their structures.
13. Rationalise the given statements and give chemical reactions:
(a) Lead(II) chloride reacts with 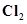 to give
to give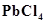 .
.
(b) Lead(IV) chloride is highly unstable towards heat.
(c) Lead is known not to form an iodide,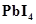 .
.
14. Suggest reasons why the B-F bond lengths in 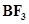 (130 pm) and
(130 pm) and 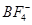 (143 pm) differ.
(143 pm) differ.
15. If B-Cl bond has a dipole moment, explain why BCl3molecule has zero dipole moment.
16. Aluminium trifluoride is insoluble in anhydrous HF but dissolves on addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous 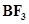 is bubbled through. Give reasons.
is bubbled through. Give reasons.
17. Suggest a reason as to why CO is poisonous.
18. How is excessive content of 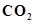 responsible for global warming?
responsible for global warming?
19. Explain structures of diborane and boric acid.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
myCBSEguide
Question Bank, Mock Tests, Exam Papers, NCERT Solutions, Sample Papers, Notes
Install Now