CBSE Question Paper 2011 class 10 Science conducted by Central Board of Secondary Education, New Delhi in the month of March 2011. CBSE previous year question papers with solution are available in myCBSEguide mobile app and cbse guide website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to cbse schools in India and abroad.
Question Paper 2011 class 10 Science

Class 10 Science list of chapters
- Chemical Reactions and Equations
- Acids, Bases and Salts
- Metals and Non-metals
- Carbon and Its Compounds
- Periodic Classification of Elements
- Life Processes
- Control and Coordination
- How do Organisms Reproduce?
- Heredity and Evolution
- Light Reflection and Refraction
- Human Eye and Colourful World
- Electricity
- Magnetic Effects of Electric Current
- Sources of Energy
- Our Environment
- Management of Natural Resources
Last Year Question Paper Class 10 Science 2011
(i)The question paper comprises of two sections, A and B. You are to attempt both the sections.
(ii) All questions are compulsory.
(iii) There is no overall choice. However internal choice has been provided in all the three questions of five marks category. Only one option in such question is to be attempted.
(iv) All questions of section A and all questions of section B are to be attempted separately.
(v) Question numbers 1 to 4 in section A are one mark questions. These are to be answered in one word or one sentence.
(vi) Question numbers 5 to 13 are two mark questions, to be answered in about 30 words each.
(vii) Question numbers 14 to 22 are three mark questions to be answered in about 50 words each.
(viii) Question numbers 23 to 25 are five mark questions, to be answered in about 70 words each.
(ix) Question numbers 26 to 41 in section B are multiple choice questions based on practical skills. Each question is a one mark question. You are to select one most appropriate response out of the four provided to you.
SECTION A
1. How many covalent bonds are there in a molecule of ethane, C H?
2. What is Tyndall effect?
3. What will happen if we kill all the organisms in one trophic level?
4. Why did United Nations act to control the production of chlorofluoro-carbons (CFCs) used in refrigerators?
(i) How do you calculate the possible valency of an element from the electronic configuration of its atoms?
(ii) Calculate the valency of an element X whose atomic number is 9.
5. How does the electronic configuration of an atom of an element relate to its position in the modem periodic table? Explain with one example.
6. How does the electronics configuration of an atom of an element relate to its position in the modern periodic table? Explain with one example.
7. State the two laws of reflection of light.
8. What is meant by the dispersion of white light? Draw a diagram to show dispersion of white light by the glass prism.
9. Explain why the planets do not twinkle but the stars twinkle.
10. Write Any two difference between binary fission and multiple fission in a tabular form as observed in cells of organisms.
11. Explain giving one example of each, the unisexual and the bisexual flowers.
12. List any four characteristics of a good fuel.
13. What are non-renewable resources of energy? Give two examples of such resources.
14. Write one chemical equation to represent each of the following types of reactions of organic substances:
(i) Esterification
(ii) Saponification
(iii) Substitution
15. Two elements X and Y belong to group 1 and 2 respectively in the same period of periodic table. Compare them with respect to:
(i) the number of valence electrons in their atoms
(ii) their valencies
(iii) metallic character
(iv) the sizes of their atoms
(v) the formulae of their oxides
(vi) the formulae of their chlorides
16. Draw the ray diagram and also state the position, the relative size and the nature of image formed by a concave mirror when the objects placed at the centre of curvature of the mirror.
17. Define ‘refractive index of a transparent medium’. What is its unit? Which has a higher refractive index, glass or water?
18. What eye defect is hypermetropia? Describe with a ray diagram how this defect of vision can be corrected by using an appropriate lens.
19. (a) List two sexually transmitted diseases in each of the following cases:
(i) Bacterial infections
(b) How may the spread of such diseases be prevented?
20. Explain with examples how the following are evidences in favour of evolution in organisms.
(i) Homologous organs
(ii) Analogous organs
(iii) Fossils
21. Explain the terms:
(i) Speciation
(ii) Natural selection
22. Explain how equal genetic contribution of male and female parents is ensured in the progeny.
23. (a) In a tabular form, differentiate between ethanol and ethanoic acid under the following heads:
(i) Physical state
(ii) Taste
(iii) NaHCO, test
(iv) Ester test
(b) Write a chemical reaction to show the dehydration of ethanol.
OR
(a) What is a soap? Why are soaps not suitable for washing clothes when the water is hard?
(b) Explain the action of soap in removing an oily spot from a piece of cloth.
24. (a) If the image formed by a lens is diminished in size and erect, for all positions of the object, what type of lens is it?
(b) Name the point on the lens through which a ray of light passes undeviated.
(c) An object is placed perpendicular to the principalaxis of a convex lens of focal length 20 cm. The distance of the object from the lens is 30 cm. Find(i) the position(ii) the magnification and(iii) the nature of the image formed.
OR
(a) One—half of a convex lens is covered with a black paper. Will such a lens produce an image of the complete object? Support your answer with a ray diagram.
(b) An object 5 cm high is held 25 cm away from a converging lens of focal length 10 cm.
(i) Draw the ray diagram.
(ii) Calculate the position and size of the image formed.
(iii) What is the nature of the image?
25. (a) Draw a diagram of the longitudinal section of a flower and label on it sepal, petal, ovary and stigma.
(b) Write the names of male and female reproductive parts of a flower.
OR
(a) What is fragmentation in organisms? Name a multicellular organism which reproduces by this method.
(b) What is regeneration in organism? Describe regeneration in Planaria with the help of a suitable diagram.
SECTION B
26. To find the focal length of a concave mirror Rahul focuses a distant object with this mirror. The chosen object should be:
(1) a tree
(2) abuilding
(3) a window
(4) the sun
27. For finding the focal length of a convex lens by obtaining the image of a distant object, one should use as the object:
(1) a well-lit distant tree
(2) window grill in the classroom
(3) any distant tree
(4) a lighted candle kept at the other end of the table
28. Mohan obtained a sharp inverted image of a distant tree on the screen placed behind the lens. He then moved the screen and tried to look through the lens in the direction of the object. He would see:
(1) a blurred image on the wall of the laboratory.
(2) an erect image of the tree on the lens.
(3) no image as the screen has been removed.
(4) an inverted image of the tree at the focus of the lens.
29. Four students A, B, C and D traced the paths of incident ray and the emergent ray by fixing pins P and Q for incident ray and pins Rand S for emergent ray fora ray of light passing through a glass slab.
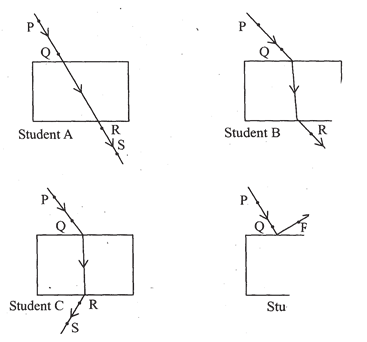
The correct emergent ray was traced by the student:
(1) A
(2) B
(3) C
(4) D
30. Rahim recorded the following sets of observations while tracing the path of a ray of light passing through a rectangular glass slab for different angles of incidence.
|
S.no. |
Angle of incidence | Angle of refraction | Angle of emergence |
|
I |
45° | 41° | 45° |
|
II |
40° | 38° | 38° |
|
III |
45° | 41° | 40° |
|
IV |
41° | 45° | 41° |
The correct observation is recorded at serial number:
(1) I
(2) II
(3) Ill
(4) IV
31. The colour of an aqueous solution of zinc sulphate as observed in the laboratory is:
(1) Green
(2) Yellow
(3) Blue
(4) Colourless
32. To show that zinc is a more active metal than copper, the correct procedure is to:
(1) add dilute nitric acid on strips of both the metals.
(2) observe transmission of heat through strips of zinc and copper.
(3) prepare solution of zinc sulphate and hang strip of copper into it.
(4) prepare solution of copper sulphate and hang strip of zinc into it.
33. Acetic acid smells like:
(1) a banana
(2) vinegar
(3) an orange
(4) a lemon
34. Acetic acid solution tums:
(1) blue litmus red
(2) Red litmus blue
(3) blue litmus colour less
(4) red litmus colour less
35. On adding NaHCO, to acetic acid, a gas is evolved which tums lime water milky due to the formation of:
(1) Calcium bicarbonate
(2) Calcium hydroxide
(3) Calcium carbonate
(4) Calcium acetate
36. A yeast cell in which budding occurs was seen to have:
(1) one bud cell
(2) two bud cells
(3) three bud cells
(4) a chain of bud cells
37. A student was given two permanent slides, one. of binary fission in amoeba and other of budding in yeast. He was asked to identify anyone difference in the nucleus of the two. One such difference, he identified correctly was:
(1) Presence of one nucleus in. amoeba, two in yeast cell and one in bud.
(2) Presence of two nuclei in centrally constricted amoeba, one in yeast cell and one in its bud.
(3) Presence of two distant nuclei in amoeba, one in yeast cell and two in bud.
(4) Presence of a single nucleus each in amoeba, yeast cell and its attached bud.
38. Binary fission is observed in which one of the following figures?

(1) A
(2) B
(3) C
(4) D
39. To determine the percentage of water absorbed by raisins, raisins are soaked in water for:
(1) 30 seconds
(2) 10 minutes
(3) 2 to 3 hours
(4) 24 hours
40. Raisins are wiped off gently before fmalweighing with help of:
(1) a filter paper
(2) a cotton piece
(3) a cloth piece
(4) a polythene piece
41. The step(s) necessary for determining the percentage of water absorbed by raisins is/are:
(1) Raisins should be completely immersed in water
(2) Raisins should be soaked in water for sufficient time
(3) Gently wipe dry the soaked raisins
(4) All of the above steps
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
CBSE Question Paper 2011 class 10 Science
Download class 10 Science question paper with solution from best CBSE App the myCBSEguide. CBSE class 10 Science paper 2011 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper.
Previous Year Question Paper for class 10 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 2009, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Mathematics Class 10
- Science Class 10 Class 10
- Social Science Class 10
- English Communicative Class 10
- English Language and Literature Class 10
- Hindi Course-A Class 10
- Hindi Course-B Class 10
- Other Subjects Class 10
To download previous year question papers for class 10 Mathematics, Science, Social Science, English Communicative, English Language and Literature, Hindi Course A, Hindi Course B, and Other subjects; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now