CBSE Question Paper 2016 class 10 Science conducted by Central Board of Secondary Education, New Delhi in the month of March 2016. CBSE previous year question papers with solution are available in myCBSEguide mobile app and cbse guide website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
Question Paper 2016 class 10 Science

Class 10 Science list of chapters
- Chemical Reactions and Equations
- Acids, Bases and Salts
- Metals and Non-metals
- Carbon and Its Compounds
- Periodic Classification of Elements
- Life Processes
- Control and Coordination
- How do Organisms Reproduce?
- Heredity and Evolution
- Light Reflection and Refraction
- Human Eye and Colourful World
- Electricity
- Magnetic Effects of Electric Current
- Sources of Energy
- Our Environment
- Management of Natural Resources
Last Year Question Paper Class 10 Science 2016
General Instructions:
1. The question paper comprises of two Sections, A and B. You are to attempt both the sections.
2. All questions are compulsory.
3. All questions of Section-A and all questions of Section-B are to be attempted separately.
4. Question numbers 1 to 3 in section-A are one mark questions.
5. Question number 4 to 6 in section-A are two marks questions.
6. Question numbers 7 to 18 in section-A are three marks questions.
7. Question number 19 to 24 in section-A are five marks questions.
8. Question number 25 to 33 in section-B are multiple choice questions based on practical skills. Each question is a one mark question. You are to select one most appropriated response our of the four provided to you.
9. Question numbers 34 to 36 in section-B are questions based on practical skills. Each question is of two marks.
Section A
Q.1 Name two components of central nervous system in Humans.
Q.2 Name the device that helps to maintain a potential difference across a conductor.
Q.3 State one use of glass cover in box type solar cookers.
Q.4 Write the chemical formula of Bleaching powder. How is bleaching powder prepared? For what purpose is it used in drinking water?
Q.5 On heating copper powder in air the surface of copper powder becomes coated with black CuO. How can this black coating be converted into brown copper? Write chemical equation for the reaction that occurs during the colour change.
Q.6 (i) Write the balanced chemical equation for the process of photosynthesis.
(ii) when do the desert plants take-up carbon dioxide and perform photosynthesis?
Q.7 Balance the following chemical equations. Write the symbols of physical states of all the reactants and the products:
(i) 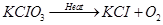
(ii) 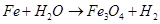
Q.8 List the raw materials needed for the manufacture of baking soda. Write the balanced chemical equation for this. What happens when baking soda is heated? Write chemical equation.
Q.9 You are given samples of three metals – sodium, magnesium, and copper. Suggest any two activities to arrange them in order of their decreasing reactivity.
Q.10 (a) Mention two observations which you will make on heating ferrous sulphate crystals in a boiling tube.
(b) On placing a zinc plate in copper sulphate solution, it was observed that the zinc plate develops holes after a few days. Give chemical equation to explain this.
(c) Silver chloride turns grey when exposed to sunlight. Give chemical equation to explain it.
Q.11 (a) state two functions of bile juice.
(b) Differentiate between the functions of enzymes pepsin and trypsin.
Q.12 Give reason to explain why endocrine glands release their secretions into the blood directly.
Q.13 Write three points of difference between photosynthesis and respiration.
Q.14 A current of 5 amperes is passed through a conductor of 12 ohms for 2 minutes. Calculate the amount of heat produced.
Q.15 (i) Will the galvanometer (G) show any defalcation when a constant current flows in coil A.
(ii) Will the galvanometer show any deflection when current in coil A is changed with the help of rheostat?
(iii) Will there be any change In deflection if the current in the coil A is changed at a faster rate? Explain your observation in each case.
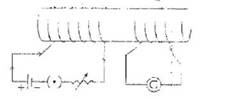
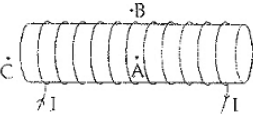
Q.16 For the current carrying Solenoid as shown below, draw magnetic field lines and giving reason explain that out of the three points A, B, and C at which point the field strength is maximum and at which point it is minimum.
Q.17 On returning home, Neha, a IXth std. Student noticed that her brother Naresh was watching T.V in the afternoon with all the lights and tans on. She noticed that the windows were closed and curtains were drawn, which made the room dark, so, Naresh had put on the lights. She calmly opened the window drew the curtains aside, which illuminated and aerated the room. Then she made Naresh put off the lights and made him understand the reason behind her action. (Assume that they are getting electric supply from Thermal Power Plant).
(a) List two values exhibited by Neha.
(b) Explain how she tried to give same values to her brother.
Q.18 List any three advantages of biogas as a source of energy.
Q.19 (a) In a tabular form write the colours of the following indicators in presence of acid and base:
Litmus solution, phenolphthalein solution, methyl orange solution.
(b) Classify the following given solutions A and B in acidic and basic, giving reason.
Solution A : 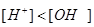
Solution B : 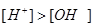
Q.20 Write the electronic configuration of magnesium (atomic no.12) and oxygen (atomic no.8) and explain the formation of magnesium oxide by electrons transfer of State the type of bond formed. Explain with reason two physical properties of compounds formed by this bonding.
Q.21 With the help of a labeled diagram explain the general scheme to illustrate how nervous impulses travel in the body.
Q.22 With the help of a circuit diagram prove that when a number of resistors are connected in series, the equivalent resistance of the combination of resistors is equal to the sum of the individual resistances of the resistors.
Calculated the equivalent resistance of the combination of three resistors of 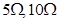 and
and 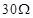 joined in parallel.
joined in parallel.
Q.23 (a) What is a magnetic field? How is the direction of magnetic field at a place determined
(b) State the rule for the direction of magnetic field produced around a current carrying straight conductor. Draw the pattern of this field.
Q.24 (a) Name the effect of electric current which is utilized in the working of an electrical fuse.
(b) How is a fuse connected in a domestic circuit?
(c) Draw a schematic labeled diagram of domestic circuit which has a provision of a main fuse, meter, one light bulb and a switch/socket.
Section B
Q.25. Remain mixed one drop of universal indicator with one drop of given solution and found that a red colour is produced, pH of the solution would be in the range of :
(a) 0 – 3
(b) 4 – 6
(c) 6 – 8
(d) 8 – 10
Q.26. Observe the following figures and choose the correct option given below:
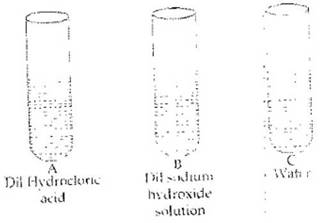
(a) pH of A, B and C are equal
(b) pH of A is greater than pH of B and C
(c) pH of C is greater than pH of A and B
(d) pH of B is greater than pH of A and C
Q.27. A student takes some zinc gargles in a test tube and adds dilute hydrochloric acid to it. He would observe that the colour of the zinc granules changes to:
(a) Brown
(b) White
(c) Yellow
(d) Black
Q.28 An iron nail was kept immersed in aluminum sulphate solution. Correct observation made after half an hour, would be.
(a) the colorless solution changed to green.
(b) a grey coating was deposited on iron nail.
(c) the iron nail became red, the colorless solution remained colorless.
(d) the solution remained colorless and no deposition observed.
Q.29 A few pieces of granulated zinc were added to copper sulphate solution in a test tube. After an hour, the colour of the solution changed form-
(a) light green to blue
(b) light green to colorless
(c) blue to light green
(d) blue to colorless
Q.30 Two resistances are connected in series as shown in the diagram.
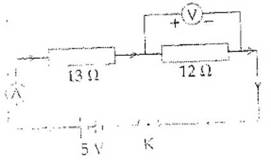
The potential difference across 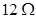 resistor will be:
resistor will be:
(a) 6 V
(b) 2.4 V
(c) 2.8 V
(d) 12 V
Q.31 A student uses a battery of adjustable voltage 0-6 V. She has to perform an experiment to determine the equivalent resistance of two resistors when connected in parallel using two resistors of value 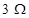 and
and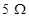 . The best choice of combination of voltmeter and ammeter to be used in the experiment is:
. The best choice of combination of voltmeter and ammeter to be used in the experiment is:
(a) Ammeter of range 0-5A and Voltmeter of range 0.10V
(b) Ammeter of range 0-5A and Voltmeter of range 0-5V
(c) Ammeter of range 0-2A and Voltmeter of range 0-10V
(d) Ammeter of range 0-5A and Voltmeter of range 0-2V
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
CBSE Question Paper 2016 class 10 Science
Download class 10 Science question paper with solution from best CBSE App the myCBSEguide. CBSE class 10 Science paper 2016 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper.
Previous Year Question Paper for class 10 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 2009, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Mathematics Class 10
- Science Class 10 Class 10
- Social Science Class 10
- English Communicative Class 10
- English Language and Literature Class 10
- Hindi Course-A Class 10
- Hindi Course-B Class 10
- Other Subjects Class 10
To download previous year question papers for class 10 Mathematics, Science, Social Science, English Communicative, English Language and Literature, Hindi Course A, Hindi Course B, and Other subjects; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now