Please explain water of crystallization.

CBSE, JEE, NEET, CUET
Question Bank, Mock Tests, Exam Papers
NCERT Solutions, Sample Papers, Notes, Videos
Posted by Sunidhi Singh 5 years, 7 months ago
- 1 answers
Related Questions
Posted by Harshal Patil 1 year, 1 month ago
- 0 answers
Posted by Pandey Ji 9 months, 1 week ago
- 0 answers
Posted by Tushar Sharma 1 year, 8 months ago
- 2 answers
Posted by Surya Kumar 1 year, 8 months ago
- 1 answers
Posted by Rani Alan 1 year, 8 months ago
- 1 answers
Posted by Ansh Preet 2 months, 4 weeks ago
- 1 answers
Posted by Harshita Tondey 1 year, 8 months ago
- 3 answers
Posted by Aadarsh Tripathi 1 year, 8 months ago
- 2 answers

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide

Gaurav Seth 5 years, 7 months ago
Water of crystallisation is a fixed number of water molecules present in one formula unit of a salt. One formula unit of copper sulphate contains five water molecules (5H20). The water molecules which form part of the structure of a crystal are called water of crystallisation. When hydrated salts are heated strongly, they lose their water of crystallisation.
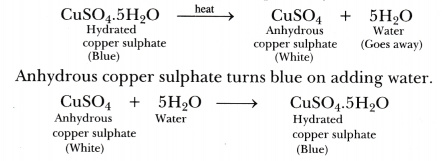
On strong heating, blue copper sulphate crystals turn white (due to the loss of water of crystallisation).
0Thank You