Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Komal Bansal 5 years, 1 month ago
- 0 answers
Posted by ☺️Vanshika Baliyan?? 5 years, 1 month ago
- 2 answers
Jass . 5 years, 1 month ago
Posted by Reena Jangra Jangra 5 years, 1 month ago
- 2 answers
Gourav Sharma 5 years, 1 month ago
Posted by Varada Menon 5 years, 1 month ago
- 1 answers
Aayan Patel 5 years, 1 month ago
Posted by Thachi Thachi 5 years, 1 month ago
- 0 answers
Posted by Thachi Thachi 5 years, 1 month ago
- 1 answers
Yogita Ingle 5 years, 1 month ago
Chemical Methods of Preparation of Colloids

Hydrophilic or Lyophobic colloidal solutions can be prepared by various chemical techniques such as:
- Double Decomposition Technique: When hydrogen sulphide is passed through a solution of arsenious oxide in distilled water, we get a colloidal solution of arsenious chloride.
As2O3 + 3H2S → As2S3 + 3H2O
- Oxidation Technique: A colloidal solution of Sulphur is made to pass through an aqueous solution of sulphur dioxide. It can also be obtained by passing the gas through a solution of an oxidization agent such as bromine water as well as nitric acid.
SO2 + 2H2S → 2H2O + 3S
H2S + [O] → H2O + S
Posted by Diksha Singh 5 years, 1 month ago
- 4 answers
Diksha Singh 5 years, 1 month ago
Aditya Singh 5 years, 1 month ago
Posted by Piyush Vaishnav 5 years, 1 month ago
- 2 answers
Posted by Ashwani Pratap Singh Lodhi 5 years, 1 month ago
- 3 answers
Himanshu Saini 5 years, 1 month ago
Posted by Krishan Kumar 5 years, 1 month ago
- 2 answers
Amrita Thakur 5 years, 1 month ago
Amrita Thakur 5 years, 1 month ago
Posted by Sachin Chandela 5 years, 1 month ago
- 1 answers
Suraj Kumar 5 years, 1 month ago
Posted by Alfaz Siddiqui 5 years, 1 month ago
- 1 answers
Mk 10 5 years, 1 month ago
Posted by Anisha Kabsuri 5 years, 1 month ago
- 1 answers
Sia ? 4 years, 8 months ago
SN1 • Polar solvent stabilizes transition state and carbocation intermediate. group. SN2 • Need polar solvent to dissolve nucleophile. Protic solvent slows rate by solvating nucleophile • Aprotic solvent increases rate by binding cation and thus freeing nucleophile.
Posted by Kirti Sharma? 5 years, 1 month ago
- 4 answers
Gourav Sharma 5 years, 1 month ago
Yogita Ingle 5 years, 1 month ago
Wurtz - Fitting reaction:
Aryl halide and alkyl halide couple in presence of sodium metal / dry ether to form alkyl benzene.
For example, bromobenzene reacts with methyl bromide in presence of sodium. dry ether to form toluene.
C6H5−Br+CH3−Br+2Na → dryether C6H5−CH3+2NaBr
Bhawna Chaudhary 5 years, 1 month ago
Posted by Ngachanyo Ps 5 years, 1 month ago
- 0 answers
Posted by Vikas Payla 5 years, 1 month ago
- 1 answers
Sanjana Dua?????❣️ 5 years, 1 month ago
Posted by Vansh Prajapati 5 years, 1 month ago
- 2 answers
Yogita Ingle 5 years, 1 month ago
A ligand is an ion or molecule, which donates a pair of electrons to the central metal atom or ion to form a coordination complex. Ambidentate ligand is a type of ligands which have the ability to bind to the central atom via the atoms of two different elements.
Examples: thiocyanate ion(NCS–) which can bind to the central metal atom or ion with either nitrogen or sulfur atoms.
Krishna Vaishist 5 years, 1 month ago
Posted by Abhi Thakur 5 years, 1 month ago
- 2 answers
Posted by Ishan Sharma 5 years, 1 month ago
- 2 answers
Posted by Vandana Patel Vandana 5 years, 1 month ago
- 0 answers
Posted by Muthu Kamatchi 5 years, 1 month ago
- 3 answers
Yogita Ingle 5 years, 1 month ago
Nitrogen with n = 2 has s and p orbitals only. If does not have d orbitals to expand its valence shell. That is why it does not form pentahalide.
Posted by Sneha Panwar 5 years, 1 month ago
- 1 answers
Bhawna Chaudhary 5 years, 1 month ago
Posted by Mr. Singh 5 years, 1 month ago
- 1 answers
Rohit Kushwaha 5 years, 1 month ago
Posted by Namitha Navaneeth Mk 5 years, 1 month ago
- 3 answers
Posted by Vishal Khandelwal 5 years, 1 month ago
- 2 answers
Yogita Ingle 5 years, 1 month ago
Benzoicacid reacts with NH3 to form ammoniumsalt which on heating decomposes to form benzamide
Ph−COOH+NH3 ⇌ Ph−COONH4+ΔPhCONH2+H2O
Posted by Vishal Khandelwal 5 years, 1 month ago
- 1 answers
Mr. Singh 5 years, 1 month ago
Posted by Vishal Khandelwal 5 years, 1 month ago
- 1 answers
Yogita Ingle 5 years, 1 month ago
Convert benzene to methyl benzene by reacting it with CH3Cl and anhydrous AlCl3. Oxidize methyl benzene with a powerful oxidizing agent like alkaline KMnO4 we will get benzoic acid.
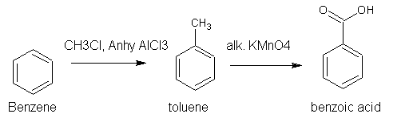
Posted by Vishal Khandelwal 5 years, 1 month ago
- 1 answers
Yogita Ingle 5 years, 1 month ago
Aniline is treated with sodium nitrite in Hydro-chloric acid at temperature 423K to form benzene diazonium chloride followed by hydrolysis by weak acid benzene .
Benzene is treated with aluminium chloride to form toulene.

Posted by Vishal Khandelwal 5 years, 1 month ago
- 2 answers
Bhawna Chaudhary 5 years, 1 month ago
Posted by Vaishali Dahiya 5 years, 1 month ago
- 1 answers
Yogita Ingle 5 years, 1 month ago
Chemisorption involves high activation energy. So, first, it increases and then decreases with increase in temperature. The initial increase is due to the heat supplied which act as activation energy required for chemisorption. But later it decreases due to the exothermic nature of adsorption at equilibrium.

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide