Molecular orbital theory of N2 and …

CBSE, JEE, NEET, CUET
Question Bank, Mock Tests, Exam Papers
NCERT Solutions, Sample Papers, Notes, Videos
Posted by Ayush Raghuwanshi 6 years, 10 months ago
- 2 answers
Gaurav Seth 5 years, 8 months ago
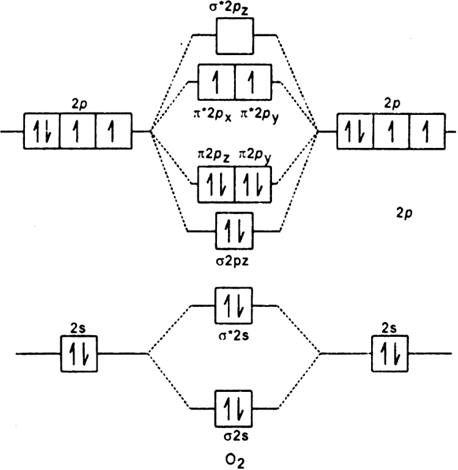
Electronic structure of oxygen atom is  Leaving out the 4 electrons in the 1s orbitals of two oxygen atoms constituting the molecule (represented as KK), the molecular orbital energy diagram for remaining 12 electrons of oxygen as molecule is shown:
Leaving out the 4 electrons in the 1s orbitals of two oxygen atoms constituting the molecule (represented as KK), the molecular orbital energy diagram for remaining 12 electrons of oxygen as molecule is shown:
Related Questions
Posted by Shaila Bombe 1 year, 5 months ago
- 1 answers
Posted by Parneet Kaur 1 year ago
- 0 answers
Posted by Rihan Mehta 1 year, 5 months ago
- 0 answers
Posted by Naman Mehra 1 year, 5 months ago
- 0 answers
Posted by "Serai✨ Wallance" 1 year, 5 months ago
- 0 answers

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide

Gaurav Seth 5 years, 8 months ago
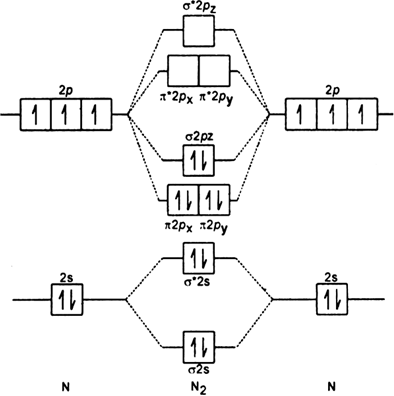
The electronic structure of nitrogen atom is Leaving out 4 electrons in the 1s orbital of two nitrogen atoms constituting the molecule (represented as KK), the molecular orbital energy diagram for remaining 10 electrons in nitrogen (N2) is as shown as below:
Leaving out 4 electrons in the 1s orbital of two nitrogen atoms constituting the molecule (represented as KK), the molecular orbital energy diagram for remaining 10 electrons in nitrogen (N2) is as shown as below:
 2px)1(σ2pz)2]
2px)1(σ2pz)2]

(i) Electronic configuration:
N2 : [KK (σ2s)2 (σ2s)2 (
(i) Bond order : Here Nh = 8 and Na = 2
The two nitrogen atoms in nitrogen molecule are linked by three covalent bonds (i.e. a triple bond).
0Thank You