
myCBSEguide App
Download the app to get CBSE Sample Papers 2024-25, NCERT Solutions (Revised), Most Important Questions, Previous Year Question Bank, Mock Tests, and Detailed Notes.
Install Now20. What happens when
(a) Borax is heated strongly,
(b) Boric acid is added to water,
(c) Aluminium is treated with dilute NaOH,
(d) 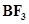 is reacted with ammonia?
is reacted with ammonia?
21. Explain the following reactions
(a) Silicon is heated with methyl chloride at high temperature in the presence of copper;
(b) Silicon dioxide is treated with hydrogen fluoride;
(c) CO is heated with ZnO;
(d) Hydrated alumina is treated with aqueous NaOH solution.
22. Give reasons:
(i) Conc. 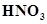 can be transported in aluminium container.
can be transported in aluminium container.
(ii) A mixture of dilute NaOH and aluminium pieces is used to open drain.
(iii) Graphite is used as lubricant.
(iv) Diamond is used as an abrasive.
(v) Aluminium alloys are used to make aircraft body.
(vi) Aluminium utensils should not be kept in water overnight.
(vii) Aluminium wire is used to make transmission cables.
23. Explain why is there a phenomenal decrease in ionisation enthalpy from carbon to silicon?
24. How would you explain the lower atomic radius of Ga as compared to Al?
25. What are allotropes? Sketch the structure of two allotropes of carbon namely diamond and graphite. What is the impact of structure on physical properties of two allotropes?
26. (a) Classify following oxides as neutral, acidic, basic or amphoteric:
CO, 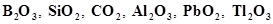
(b) Write suitable chemical equations to show their nature.
27. In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
28. When metal X is treated with sodium hydroxide, a white precipitate (A) is obtained, which is soluble in excess of NaOH to give soluble complex (B). Compound (A) is soluble in dilute HCl to form compound (C). The compound (A) when heated strongly gives (D), which is used to extract metal. Identify (X), (A), (B), (C) and (D). Write suitable equations to support their identities.
29. What do you understand by (a) inert pair effect (b) allotropy and(c) catenation?
30. A certain salt X, gives the following results.
(i) Its aqueous solution is alkaline to litmus.
(ii) It swells up to a glassy material Y on strong heating.
(iii) When conc. 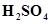 is added to a hot solution of X, white crystal of an acid Z separates out.
is added to a hot solution of X, white crystal of an acid Z separates out.
Write equations for all the above reactions and identify X, Y and Z.
31. Write balanced equations for:
(i) 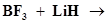
(ii) 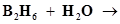
(iii) 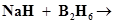
(iv) 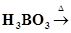
(v) 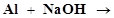
(vi) 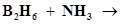
32. Give one method for industrial preparation and one for laboratory preparation of CO and 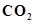 each.
each.
33. An aqueous solution of borax is
(a) neutral
(b) amphoteric
(c) basic
(d) acidic
34. Boric acid is polymeric due to
(a) its acidic nature
(b) the presence of hydrogen bonds
(c) its monobasic nature
(d) its geometry
35. The type of hybridisation of boron in diborane is
(a) sp (b) 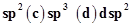
36. Thermodynamically the most stable form of carbon is
(a) diamond
(b) graphite
(c) fullerenes
(d) coal
37. Elements of group 14
(a) exhibit oxidation state of +4 only
(b) exhibit oxidation state of +2 and +4
(c) form 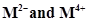 ion
ion
(d) form 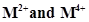 ions
ions
38. If the starting material for the manufacture of silicones is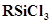 , write the structure of the product formed.
, write the structure of the product formed.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
myCBSEguide
Question Bank, Mock Tests, Exam Papers, NCERT Solutions, Sample Papers, Notes
Install Now