Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Dora Moon 2 years, 11 months ago
- 1 answers
Posted by Ramandeep Chauhan 2 years, 11 months ago
- 0 answers
Posted by Tanushree Deb Burman 2 years, 11 months ago
- 1 answers
Preeti Dabral 2 years, 11 months ago
Order of a reaction is the sum of the coefficients of the reacting species involved in the rate equation. Molecularity is the number of reacting species involved in simultaneous collisions in an elementary or simplest reaction.
Posted by Tushar Goswami 3 years ago
- 1 answers
Posted by Shanaya Rajput 3 years ago
- 1 answers
Anjan Karthi 3 years ago
Posted by Abhay Choudhary 3 years ago
- 1 answers
Shanaya Rajput 3 years ago
Posted by Jyoti Shinde 3 years ago
- 2 answers
Satyam Pratap 3 years ago
Na. Shakthi 3 years ago
Posted by Tushar Goswami 3 years ago
- 2 answers
Abhishek Meena Saxena 3 years ago
Few rules to keep homework help section safe, clean and informative.
- Don't post personal information, mobile numbers and other details.
- Don't use this platform for chatting, social networking and making friends. This platform is meant only for asking subject specific and study related questions.
- Be nice and polite and avoid rude and abusive language. Avoid inappropriate language and attention, vulgar terms and anything sexually suggestive. Avoid harassment and bullying.
- Ask specific question which are clear and concise.
Remember the goal of this website is to share knowledge and learn from each other. Ask questions and help others by answering questions.
Na. Shakthi 3 years ago
Posted by Jyoti Shinde 3 years ago
- 2 answers
Na. Shakthi 3 years ago
Tushar Goswami 3 years ago
Posted by Malik Ujaiv 3 years ago
- 2 answers
Punya Yadav 3 years ago
Few rules to keep homework help section safe, clean and informative.
- Don't post personal information, mobile numbers and other details.
- Don't use this platform for chatting, social networking and making friends. This platform is meant only for asking subject specific and study related questions.
- Be nice and polite and avoid rude and abusive language. Avoid inappropriate language and attention, vulgar terms and anything sexually suggestive. Avoid harassment and bullying.
- Ask specific question which are clear and concise.
Remember the goal of this website is to share knowledge and learn from each other. Ask questions and help others by answering questions.
Posted by Malik Ujaiv 3 years ago
- 1 answers
Diana Chaudhary 3 years ago
Posted by Abhishek Patel 3 years ago
- 1 answers
Posted by Tanay Naik 3 years ago
- 2 answers
Posted by Aarzoo Saini 3 years ago
- 1 answers
Posted by Jyoti Shinde 3 years ago
- 5 answers
Bava Samyuka 3 years ago
Suryansh Srivastav 3 years ago
Posted by Tushar Goswami 3 years ago
- 3 answers
Jyoti Shinde 3 years ago
Posted by Mohsin Khan 3 years ago
- 3 answers
Muskan Garg 2 years, 11 months ago
Rana Sidhu 3 years ago
Rana Sidhu 3 years ago
Posted by Tushar Goswami 3 years ago
- 3 answers
Jatin Grewal 3 years ago
Posted by Arpit Gupta 3 years ago
- 1 answers
Posted by Pratibha Marmat 3 years ago
- 2 answers
Na. Shakthi 3 years ago
Posted by Vishakha Mogha 3 years ago
- 2 answers
Posted by Jhalak Aggarwal 3 years ago
- 1 answers
Srija Srija 3 years ago
Posted by Champa Gupta 3 years ago
- 1 answers
Posted by Shailja Singh 2 years, 11 months ago
- 1 answers
Preeti Dabral 2 years, 11 months ago
{tex}\alpha {/tex} -glucose and {tex}\beta {/tex} -glucose differ in configuration at C-1. They Differ in optical rotation. The differ in melting point also. Such isomers differing in configuration at C-1 are called anomers.
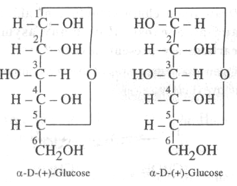
Posted by Shailja Singh 3 years ago
- 1 answers
Posted by Divya. Kumari 3 years ago
- 5 answers
Posted by Sangita Boruah 3 years ago
- 4 answers
Na. Shakthi 3 years ago
Ankush Bishnoi 3 years ago
Karan Gadhekar 3 years ago
Few rules to keep homework help section safe, clean and informative.
- Don't post personal information, mobile numbers and other details.
- Don't use this platform for chatting, social networking and making friends. This platform is meant only for asking subject specific and study related questions.
- Be nice and polite and avoid rude and abusive language. Avoid inappropriate language and attention, vulgar terms and anything sexually suggestive. Avoid harassment and bullying.
- Ask specific question which are clear and concise.
Remember the goal of this website is to share knowledge and learn from each other. Ask questions and help others by answering questions.
Rashi Nain 3 years ago
Posted by Nandani Sharma 3 years ago
- 2 answers
Sania Rahman 3 years ago
Sania Rahman 3 years ago

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide
..... ..... 2 years, 11 months ago
0Thank You