Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Abhinav Tripathi 7 years ago
- 1 answers
Posted by Govind Kumar 7 years ago
- 1 answers
Gaurav Seth 7 years ago
According to Bohr's theory:
(i) The atom consists of a small (positively charged) nucleus at its centre.
(ii) The whole mass of the atom is concentrated in the nucleus and the volume of the nucleus is smaller than the volume of the atom by a ratio of about 1 : 105.
(iii) All the protons and neutrons of the atom are contained in the nucleus.

(iv) The electrons of the atom revolve around the nucleus in definite circular paths known as orbits or which are designated as K, L, M, N etc. or numbered as (n) = 1, 2, 3, 4 etc. outward from the nucleus.
(v) Each orbit is associated with a fixed amount of energy. Therefore, these orbits are also known as energy levels or energy shells.
(vi) The energy of the atom changes when an electron jumps from one state (energy level) to another state (energy level). As long as an electron remains in a particular orbit, it does not lose or gain energy.
Posted by Prakhar J 7 years ago
- 0 answers
Posted by Pankaj Narvariya 7 years ago
- 0 answers
Posted by Swayam Phutela 7 years ago
- 1 answers
Gaurav Yadav 7 years ago
Posted by Abhishek Khare 7 years ago
- 1 answers
Yogita Ingle 7 years ago
Depression of freezing point
- Freezing point of a substance is the temperature at which the vapour pressure
of the substance in its liquid phase is equal to the vapour pressure in the solid phase. - If the vapour pressure of the solution is equal to the vapour pressure of the pure solid solvent then the solution freezes.
- According to Raoult’s law, addition of non-volatile solute decreases the vapour pressure of the solvent and would be equal to that of solid solvent at lower This decreases the freezing point of thesolvent.
- The decrease in the freezing point = ΔTf = Tf – Tf0. This is known as depression in freezing point.
Tf0 = freezing point of pure solvent
Tf0 = freezing point when non-volatile solute is dissolved
Posted by Shruti Binay 7 years ago
- 1 answers
Posted by Ishita Chatterjee 7 years ago
- 1 answers
Posted by Aman Singh 7 years ago
- 1 answers
Adrija Pal ? 7 years ago
Posted by Azhan Khan 7 years ago
- 1 answers
Gaurav Seth 7 years ago
Zone refining: The method is based on the principle that the impurities are more soluble in the melt than in the solid state of the metal. A circular mobile heater is fixed at one end of a rod of impure metal. The molten zone moves along with the heater which is moved forward.
Posted by Shaurya Omar 7 years ago
- 2 answers
Shaurya Omar 7 years ago
Posted by Abijith Paul George 7 years ago
- 2 answers
Posted by Saima Jain 7 years ago
- 2 answers
Saima Jain 7 years ago
Posted by Gurnoor Padda 7 years ago
- 2 answers
G K 7 years ago
Posted by Jitendra Kumar 7 years ago
- 2 answers
Adrija Pal ? 7 years ago
Posted by Prachi Prabha 7 years ago
- 0 answers
Posted by Ayesha Firdous 7 years ago
- 1 answers
Gaurav Seth 7 years ago
A mixture of equal amounts of two enantiomers is known as racemic mixture. For example: When a 3° halide undergoes substitution with KOH, the reaction proceeds through SN 1 mechanism forming the racemic mixture in which one of the products has the same configuration as a reactant, while the other product has an inverted configuration.
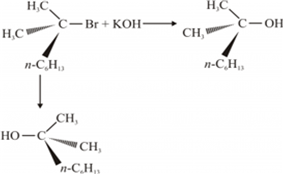
Posted by Divyansh Rathore 7 years ago
- 1 answers
Madhu Wwe 7 years ago
Posted by Arnav Sharma 7 years ago
- 0 answers
Posted by Deign Immanual 4 years, 9 months ago
- 1 answers
Posted by Mandvi Gupta 7 years ago
- 1 answers
Prashant Rawat 7 years ago
Posted by Akash Joshi 7 years ago
- 0 answers
Posted by Ashwini Kumar Mohanty 7 years ago
- 1 answers
Posted by Vinar Yadav 7 years ago
- 0 answers
Posted by Jewel Maria 7 years ago
- 1 answers
Posted by Abhaysingh Singh Yadav 7 years ago
- 0 answers
Posted by Appy Garnara 7 years ago
- 1 answers
Yogita Ingle 7 years ago
Solids have strong intermolecular forces of attraction. The constituent particles of solids cannot move from their positions they can only vibrate from their mean positive. That is why do solids have a definite volume.

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide

Ayush Verma 7 years ago
1Thank You