Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Kenpanna Konganoli Konganolli 4 years, 4 months ago
- 1 answers
Posted by Pk . 4 years, 4 months ago
- 2 answers
?Tamanna ?? 4 years, 3 months ago
Posted by Harsh Sharma 4 years, 4 months ago
- 2 answers
Sia ? 4 years, 4 months ago
Interhalogen compounds are more reactive than their parent halogen. This is because the bond between two different halogen atoms is weaker than the bond between the same halogen atoms.
Rimmi Bhardwaj 4 years, 4 months ago
Posted by Lovejeet Brar 4 years, 4 months ago
- 1 answers
Posted by Jyoti Sharma 4 years, 4 months ago
- 1 answers
Garima Yadav 4 years, 4 months ago
Posted by Deepak Patel 4 years, 4 months ago
- 1 answers
Posted by Saksham Mittal 4 years, 4 months ago
- 4 answers
Posted by Atharva Verma 4 years, 4 months ago
- 2 answers
Sia ? 4 years, 4 months ago
The Conversion reaction of 1 Chlorobutanol to 1 butanol takes place under a Strong Base like KOH. The 1 Chlorobutanol reacts with KOH to Give butane-1-Ol as the Desired product along with HCl as a by-product.
Simran Kumari 4 years, 4 months ago
Posted by Aditi Choudhary 4 years, 4 months ago
- 1 answers
Sia ? 4 years, 4 months ago
| Hypotonic solution | Hypertonic solution |
| The solution outside the cell has a higher soluble concentration than inside the cell. Thus, the water molecules move outside the cell from inside. | The solution outside the cell has a less concentrated solution than inside the cell. Thus, the water molecules move inside the cell from outside. |
| A solution whose concentration is less than the cell sap or inside of a cell. | A solution whose concentration is more than the cell sap or that inside a cell. |
| A plant cell becomes turgid when putting in a hypotonic solution. | A plant cell undergoes plasmolysis in a hypertonic solution. |
Posted by Sharveshnii Mohanraj 4 years, 4 months ago
- 1 answers
Posted by Priyadarshan Singh 4 years, 4 months ago
- 1 answers
Posted by Calvin Bhalerao 4 years, 4 months ago
- 1 answers
Sia ? 4 years, 4 months ago
T = {tex}{37^0}C{/tex} = 310 k
{tex}\pi = {\rm{ }}CRT{/tex} {tex} = \frac{n}{v}RT{/tex}
{tex} = 0.25 \times 0.083 \times 310{/tex}
= 6.43 bar.
Posted by Kajal Pal 4 years, 4 months ago
- 4 answers
Aseem Mahajan 4 years, 4 months ago
Posted by Shrestha Pal 4 years, 4 months ago
- 2 answers
Aseem Mahajan 4 years, 4 months ago
Posted by Muhsina ❤ 4 years, 4 months ago
- 1 answers
Posted by Hari Haran 4 years, 5 months ago
- 2 answers
Rohit Kumar 4 years, 4 months ago
Posted by 🤟Royal Thakur 🤟 4 years, 5 months ago
- 2 answers
Aseem Mahajan 4 years, 5 months ago
Confirm carefully about that khushi kamboj I highly doubt its him
Posted by Arunav Chatterjee 4 years, 5 months ago
- 1 answers
Posted by Arman Dwibedy 4 years, 5 months ago
- 2 answers
Dishant Rajput 4 years, 3 months ago
Posted by Aseem Mahajan 4 years, 5 months ago
- 5 answers
Aseem Mahajan 4 years, 5 months ago
Posted by Mansi Sagar 4 years, 5 months ago
- 1 answers
Aseem Mahajan 4 years, 5 months ago
Butan-2-ol has OH group on carbin bext to that of terminal carbon of n-butyl chain
Posted by Arunav Chatterjee 4 years, 5 months ago
- 1 answers
Itz Kaur 4 years, 5 months ago
Posted by Hephzibah Jacob 4 years, 5 months ago
- 3 answers
Posted by Nishita Talukdar 4 years, 5 months ago
- 1 answers
Garima Yadav 4 years, 4 months ago
Posted by Anuj Kumar 4 years, 5 months ago
- 2 answers
🤟Royal Thakur 🤟 4 years, 5 months ago
Posted by Khushali Patharkar 4 years, 5 months ago
- 3 answers
Parvez Rasool 4 years, 5 months ago
Posted by Tania J 4 years, 5 months ago
- 1 answers
Posted by 🤟Royal Thakur 🤟 4 years, 5 months ago
- 4 answers
Shashwat Singh 4 years, 5 months ago
Posted by Sudhanshu Singh 4 years, 5 months ago
- 2 answers
Suryansh Shrivastava 4 years, 4 months ago
Devika Tiwari 4 years, 5 months ago

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide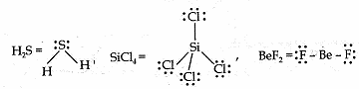
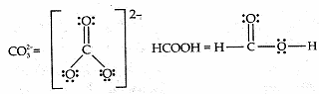

Sia ? 4 years, 4 months ago
Defects or Imperfections in crystalline solid can be divided into four groups namely line defects, point defects, volume defects and surface defects.
0Thank You