Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Sarthak Singh 7 years, 1 month ago
- 1 answers
Posted by Bishal Rawat 7 years, 1 month ago
- 0 answers
Posted by Kartik Tanwar 7 years, 1 month ago
- 2 answers
Keerthu Raghu 7 years, 1 month ago
Gursharn Kaur 7 years, 1 month ago
Posted by Rutuparnna Mangal 7 years, 1 month ago
- 0 answers
Posted by Mann Patidar 7 years, 1 month ago
- 1 answers
Sahil Sharma 7 years, 1 month ago
Posted by Madhup Kumar 7 years, 1 month ago
- 1 answers
Gaurav Seth 7 years, 1 month ago
The radius of gyration of a body about a given axis is the perpendicular distance of a point P from the axis, where if whole mass of the body were concentrated, as the body shall have the same moment of inertia as it has with the actual distribution of mass.
Radius of gyration is represented by K.
Posted by Madhup Kumar 7 years, 1 month ago
- 1 answers
Gaurav Seth 7 years, 1 month ago
Consider a liquid drop of radius R and surface tension T.
Due to surface tension the molecules on the surface film experience the net force in inward direction normal to the surface.
Therefore there is more pressure inside than outside.
Let pi be the pressure inside the liquid drop and po be the pressure outside the drop.
Therefore excess of pressure inside the liquid drop is,
p = p1– p0
Due to excess of pressure inside the liquid drop the free surface of the drop will experience the net force in outward direction due to which the drop will expand.
Let the free surface displace by dR under isothermal conditions.
Therefore excess of pressure does the work in displacing the surface and that work will be stored in the form of potential energy.
The work done by excess of pressure in displacing the surface is,
dW = Force x displacement
= (Excess of pressure x surface area) x displacement of surface

Increase in the potential energy is,
dU = surface tension x increase in area of the free surface

The above expression gives us the pressure inside a liquid drop.
Posted by Madhup Kumar 7 years, 1 month ago
- 0 answers
Posted by Madhup Kumar 7 years, 1 month ago
- 1 answers
Gaurav Seth 7 years, 1 month ago
Derive an expression for work done in isothermal process
<section data-topic-id="6324" id="topic"> <article data-post-id="7452" data-topic-id="6324" data-user-id="2" id="post_1">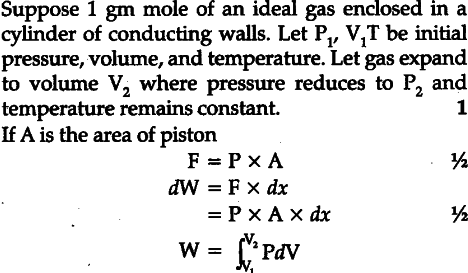
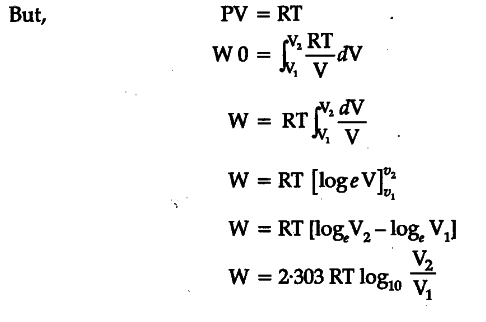
Posted by Madhup Kumar 7 years, 1 month ago
- 1 answers
Gaurav Seth 7 years, 1 month ago
Statement: For the streamline flow of non-viscous and incompressible liquid, the sum of potential energy, kinetic energy and pressure energy is constant.


Proof:
Let us consider the ideal liquid of density ρ flowing through the pipe LM of varying cross-section.
Let P1 and P2 be the pressures at ends L and M and A1 and A2 be the areas of cross-sections at ends L and M respectively.
Let the liquid enter with velocity V1 and leave with velocity v2.
Let A1 > A2.
Now, by equation of continuity,

Since, A1 > A2
Therefore,
v2 > v1 and P1 > P2
Let, m be the mass of liquid enetring at end L in time t.
The liquid will cover a distance = v1t
Therefore, the work done by pressure on the liquid at end L in time t is,
W1 = Force 
= P1A1v1t ... (1)
Since same mass m leaves the pipe at end M in same time t, in which liquid will cover the distance given by v2t.
Therefore, work done by liquid against the force due to pressure P1 is given by,
W2 = P2A2v2t ... (2)
Net ork done by pressure on the liquid in time t is,
W = W1 - W2
=P1A1v1t - P2A2v2t ... (3)
This work done on liquid by pressure increases it's kinetic energy and potential energy.
Increase in K.E of liquid is,

That is,

Posted by Abhi Kohal Rajput 7 years, 1 month ago
- 2 answers
Sanidhya Jadaun? 7 years, 1 month ago
Sanidhya Jadaun? 7 years, 1 month ago
Posted by Chandan ??? 7 years, 1 month ago
- 1 answers
Anil Kumar 7 years, 1 month ago
Posted by Harish Kumar 7 years, 1 month ago
- 0 answers
Posted by Annu Sharma 7 years, 1 month ago
- 0 answers
Posted by Varsha Khatri 7 years, 1 month ago
- 1 answers
Sanidhya Jadaun? 7 years, 1 month ago
Posted by Nikita Bansal 7 years, 1 month ago
- 1 answers
Priya Dharshini ? 7 years, 1 month ago
Posted by Nikita Parakh 7 years, 1 month ago
- 2 answers
Yogita Ingle 7 years, 1 month ago
i) It is an orderly type of motion in which the liquid flows in parallel layers while, turbulent motion is disorderly type of motion.
ii) Every particle of the liquid follows the path of it's preceeding particle and travel with the same velocity in magnitude and direction whereas, the motion of particles of the liquids becomes different at different points in turbulent flow.
iii) The velocity of streamline flow is less than critical velocity but, in turbulent motion the liquid moves with a velocity greater than the critical velocity of the liquid.
iv) Streamline flow is laminar whereas, turbulent flow is non-laminar.
Sanidhya Jadaun? 7 years, 1 month ago
Posted by Himanshu Singh 7 years, 1 month ago
- 0 answers
Posted by Aman Garg 7 years, 1 month ago
- 1 answers
Yogita Ingle 7 years, 1 month ago
STP : Standard temperature and pressure, abbreviated STP, refers to nominal conditions in the atmosphere at sea level. This value is important to physicists, chemists, engineers, and pilots and navigators.
Standard temperature is defined as zero degrees Celsius (0 0C), which translates to 32 degrees Fahrenheit (32 0F) or 273.15 degrees kelvin (273.15 0K). This is essentially the freezing point of pure water at sea level, in air at standard pressure.
NTP : Normal Temperature and Pressure - is defined as air at 20oC (293.15 K, 68oF) and 1 atm ( 101.325 kN/m2, 101.325 kPa, 14.7 psia, 0 psig, 29.92 in Hg, 760 torr). Density 1.204 kg/m3 (0.075 pounds per cubic foot
Posted by Kapil Kumar 7 years, 1 month ago
- 2 answers
Rahul Gond 7 years, 1 month ago
Jawahharlal Bandey 7 years, 1 month ago
Posted by Richa Purvey 7 years, 1 month ago
- 0 answers

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide
Rohit Gurav 7 years, 1 month ago
2Thank You