Ask questions which are clear, concise and easy to understand.
Ask QuestionPosted by Reena Rajeev 4 years, 5 months ago
- 0 answers
Posted by Utkarsh Singh 4 years, 5 months ago
- 3 answers
Rishabh Roy 3 years, 7 months ago
Posted by Sri Vineetha 4 years, 6 months ago
- 0 answers
Posted by Rohit Kumar Jha 4 years, 6 months ago
- 0 answers
Posted by Gautam Pandey 4 years, 7 months ago
- 0 answers
Posted by Vinay Tiwari 4 years, 7 months ago
- 1 answers
Posted by Arshi Khan 4 years, 7 months ago
- 0 answers
Posted by Prince Kumar Tiwari 4 years, 7 months ago
- 0 answers
Posted by Mohammed Faisal 4 years, 7 months ago
- 1 answers
Posted by Shaik Mohameed 4 years, 7 months ago
- 1 answers
Posted by Rimeki Lyngdoh Mawphlang 4 years, 8 months ago
- 2 answers
Deepak Gupta 4 years, 7 months ago
Posted by Himansu Behera 4 years, 8 months ago
- 2 answers
Monika Chauhan 4 years, 8 months ago
Yogita Ingle 4 years, 8 months ago
The energy required to remove an electron from a neutral isolated gaseous stom and convert it into a positively charged gaseous ion is called ionisation energy or first ionisation energy.
Ionisation energy required to remove the second electron from a monovalent cation is called second ionization energy.
Similarly, the energy required to remove the third electron from a divalent cation is called the third ionisation energy.
Posted by Sachin Master 4 years, 9 months ago
- 1 answers
Posted by Basam Bhavya 4 years, 9 months ago
- 1 answers
Posted by Kartik Kitukale 4 years, 10 months ago
- 0 answers
Posted by Bharat Maurya 4 years, 10 months ago
- 1 answers
Posted by Parvesh Sriram 4 years, 10 months ago
- 0 answers
Posted by Md. Mustafa Khan 4 years, 11 months ago
- 0 answers
Posted by Abhushek Vishwakarma 4 years, 11 months ago
- 3 answers
Posted by Md. Mustafa Khan 2 years, 9 months ago
- 1 answers
Posted by Omkar Jadhav 4 years, 11 months ago
- 1 answers
Bikram Parui 4 years, 11 months ago
Posted by Abhishek Kashyap 5 years ago
- 1 answers
Posted by Ankit Dubey 5 years ago
- 3 answers
Posted by Shivangi Yadav 5 years, 1 month ago
- 3 answers
Posted by Shivangi Yadav 5 years, 1 month ago
- 2 answers
Gaurav Seth 5 years, 1 month ago
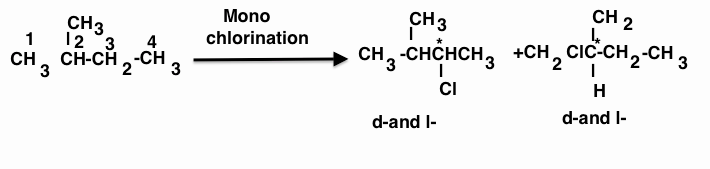
Chlorination at C-2 and C-4 produces no chiral compound.
Hence (b) is the correct answer.
Posted by Shivangi Yadav 5 years, 1 month ago
- 1 answers
Gaurav Seth 5 years, 1 month ago
Among the following compounds, the most acidic is: [JEE-2011]
-
a)
p - nitrophenol
-
b)
p - hydroxybenzoic acid
-
c)
o - hydroxybenzoic acid
-
d)
p - toluic acid
Correct answer is option 'C'.
Ortho-isomer, a strong hydrogen bridge can be formed between the phenolic -OH and the the carboxylate anion. This will tend to stabilize the conjugated carboxylate base, and hence will increase acidity.
this is the order of acidic
ortho hydroxy B.acid > meta hydroxy B.acid > benzoic acid > para hydroxy B. acids.

myCBSEguide
Trusted by 1 Crore+ Students

Test Generator
Create papers online. It's FREE.

CUET Mock Tests
75,000+ questions to practice only on myCBSEguide app
 myCBSEguide
myCBSEguide
