CBSE Chemistry Chapter 1 Some Basic Concepts of Chemistry class 11 Notes Chemistry in PDF are available for free download in myCBSEguide mobile app. The best app for CBSE students now provides Some Basic Concepts of Chemistry class 11 Notes Chemistry latest chapter wise notes for quick preparation of CBSE board exams and school based annual examinations. Class 11 Chemistry notes on Chapter 1 Some Basic Concepts of Chemistry class 11 Notes Chemistry are also available for download in CBSE Guide website.
CBSE Guide Some Basic Concepts of Chemistry class 11 Notes
CBSE guide notes are the comprehensive notes which covers the latest syllabus of CBSE and NCERT. It includes all the topics given in NCERT class 11 Chemistry text book. Users can download CBSE guide quick revision notes from myCBSEguide mobile app and my CBSE guide website.
Some Basic Concepts of Chemistry class 11 Notes Chemistry
Download CBSE class 11th revision notes for Chapter 1 Some Basic Concepts of Chemistry class 11 Notes Chemistry in PDF format for free. Download revision notes for Some Basic Concepts of Chemistry class 11 Notes Chemistry and score high in exams. These are the Some Basic Concepts of Chemistry class 11 Notes Chemistry prepared by team of expert teachers. The revision notes help you revise the whole chapter in minutes. Revising notes in exam days is on of the best tips recommended by teachers during exam days.
Download Revision Notes as PDF
CBSE Class 11 Chemistry
Revision Notes
Chapter 1
Some Basic Concepts of Chemistry
- Importance and Scope of Chemistry
- Nature of Matter
- Laws of Chemical Combinations
- Dalton’s Atomic Theory
- Atomic and Molecular Masses
- Stoichiometry and Stoichiometric Calculations
Some Important Points and Terms of the Chapter
1. Anything which has mass and occupies space is called matter.
2. Matters exist in three physical states viz. solid, liquid and gas.
3. In solids, these particles are held very close to each other in an orderly fashion and there is not much freedom of movement. In liquids, the particles are close to each other but they can move around. However, in gases, the particles are far apart as compared to those present in solid or liquid states and their movement is easy and fast.
4. Solids have definite volume and definite shape.
5. Liquids have definite volume but not the definite shape. They take the shape of the container in which they are placed.
6. Gases have neither definite volume nor definite shape. They completely occupy the container in which they are placed.
7. A mixture contains two or more substances present in it (in any ratio) which are called its components.
8. A mixture may be homogeneous or heterogeneous.
9. In a homogeneous mixture, the components completely mix with each other and its composition is uniform throughout. Sugar solution and air are thus, the examples of homogeneous mixtures.
10. In heterogeneous mixtures, the composition is not uniform throughout and sometimes the different components can be observed. For example, the mixtures of salt and sugar, grains and pulses along with some dirt (often stone) pieces, are heterogeneous mixtures..
11. The components of a mixture can be separated by using physical methods such as simple hand picking, filtration, crystallization, distillation etc.
12. Pure substances have characteristics different from the mixtures. They have fixed composition, Copper, silver, gold, water, glucose are some examples of pure substances. Glucose contains carbon, hydrogen and oxygen in a fixed ratio and thus, like all other pure substances has a fixed composition. Also, the constituents of pure substances cannot be separated by simple physical methods.
13. An element consists of only one type of particles. These particles may be atoms or molecules. Sodium, copper, silver, hydrogen, oxygen etc. are some examples of elements. They all contain atoms of one type. However, the atoms of different elements are different in nature. Some elements such as sodium or copper, contain single atoms held together as their constituent particles whereas in some others, two or more atoms combine to give molecules of the element. Thus, hydrogen, nitrogen and oxygen gases consist of molecules in which two atoms combine to give their respective molecules.
14. When two or more atoms of different elements combine, the molecule of a compound is obtained. The examples of some compounds are water, ammonia, carbon dioxide, sugar etc. the atoms of different elements are present in a compound in a fixed and definite ratio and this ratio is characteristic of a particular compound.
15. The SI system (Systeme International d Unités –abbreviated as SI)) has seven base units and they are listed in
Table 1.1 Base Physical Quantities and their Unites
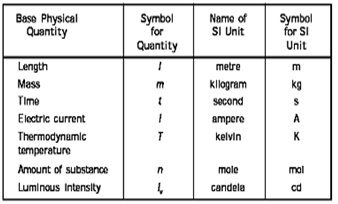
16. Mass of a substance is the amount of matter present in it while weight is the force exerted by gravity on an object. The mass of a substance is constant whereas its weight may vary from one place to another due to change in gravity.
17. Volume has the units of . So in SI system, volume has units of . A common unit, litre (L) which is not an SI unit, is used for measurement of volume of liquids.
18. Density of a substance is its amount of mass per unit volume SI units of density kg
This unit is quite large and a chemist often expresses density in .
19. There are three common scales to measure temperature Here, K is the SI unit.
20. The Kelvin scale is related to Celsius scale as follows:
21. The °F scale is related to Celsius scale as follows
22. In scientific notation (exponential Notation) any number can be represented in the form where n is an exponent having positive or negative values and N can vary between 1 to 10. Thus, we can write in scientific notation. Note that while writing it, the decimal had to be moved to the left by two places and same is the exponent (2) of 10 in the scientific notation. Similarly, 0.00016 can be written as . Here the decimal has to be moved four places to the right and (– 4) is the exponent in the scientific notation.
23. In Significant figures are meaningful digits which are known with certainty. The uncertainty is indicated by writing the certain digits and the last uncertain digit. Thus, if we write a result as 11.2 mL, we say the 11 is certain and 2 is uncertain and the uncertainty would be 1 in the last digit. Unless otherwise stated, an uncertainty of +1 in the last digit is always understood.
24. There are certain rules for determining the number of significant figures. These are stated below:
a) All non-zero digits are significant. For example in 285 cm, there are three significant figures and in 0.25 mL, there are two significant figures.
b) Zeros preceding to first non-zero digit are not significant. Such zero indicates the position of decimal point. Thus, 0.03 has one significant figure and 0.0052 has two significant figures.
c) Zeros between two non-zero digits are significant. Thus, 2.005 has four significant figures.
d) Zeros at the end or right of a number are significant provided they are on the right side of the decimal point. For example, 0.200 g has three significant figures. But, if otherwise, the terminal zeros are not significant if there is no decimal point. For example, 100 has only one significant figure, but 100. has three significant figures and 100.0 has four significant figures. Such numbers are better represented in scientific notation. We can express the number for one significant figure for two significant figures and for three significant figures.
e) Counting numbers of objects, for example, 2 balls or 20 eggs, have infinite significant figures as these are exact numbers and can be represented by writing infinite number of zeros after placing a decimal i.e.,2 = 2.000000 or 20 = 20.000000
f) In numbers written in scientific notation, all digits are significant e.g has three significant figures, and has four significant figures.
25. Law of Conservation of Mass. Antoine Lavoisier established the Law of Conservation of Mass. It states that matter can neither be created nor destroyed.
26. Law of Definite Proportions states that a given compound always contains exactly the same proportion of elements by weight.
27. Law of Multiple Proportions states that if two elements can combine to form more than one compound, the masses of one element that combine with a fixed mass of the other element, are in the ratio of small whole numbers.
28. Gay Lussac’s Law of Gaseous Volumes: This law was given by Gay Lussac in 1808. He observed that when gases combine or are produced in a chemical reaction they do so in a simple ratio by volume provided all gases are at same temperature and pressure.
29. In 1811, Avogadro proposed that equal volumes of gases at the same temperature and pressure should contain equal number of molecules.
30. In 1808, Dalton published ‘A New System of Chemical Philosophy‘ in which he proposed the following :
a) Matter consists of indivisible atoms.
b) All the atoms of a given element have identical properties including identical mass. Atoms of different elements differ in mass.
c) Compounds are formed when atoms of different elements combine in a fixed ratio.
d) Chemical reactions involve reorganization of atoms. These are neither created nor destroyed in a chemical reaction.
e) Dalton‘s theory could explain the laws of chemical combination.
31. One atomic mass unit is defined as a mass exactly equal to one twelfth the mass of one carbon – 12 atom
32. Molecular mass is the sum of atomic masses of the elements present in a molecule. It is obtained by multiplying the atomic mass of each element by the number of its atoms and addingthem together. Molecular mass expressed in grams is called gram molecular mass
Formula Mass: Sum of atomic masses of all atoms in a formula unit of the compound
33. The mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 0.012 kilogram of carbon-12; its symbol is -mol.When the mole is used, the elementary entities must be specified and may be atoms, molecules, ions, electrons, other particles, or specified groups of such particles. This number of entities in 1 mol is so important that it is given a separate name and symbol. It is known as ‘Avogadro constant‘, denoted by in honor of Amedeo Avogadro.
34. An empirical formula represents the simplest whole number ratio of various atoms present in a compound whereas the molecular formula shows the exact number of different types of atoms present in a molecule of a compound.
Molecular formula shows the exact number of different types of atoms present in a molecule of a compound. If the mass per cent of various elements present in a compound is
known, its empirical formula can be determined.
Molecular formula = n (Empirical formula) , where n is a simple number and may have values 1, 2, 3….
Following steps should be followed to determine empirical formula of the compound:
Step 1: Conversion of mass per cent of various elements into grams.
Step 2: Convert mass obtained into number of moles.
Step 3: Divide the mole value obtained by the smallest mole value (out of the mole value of various elements calculated)
Step 4: If the ratios are not whole numbers, then they may be converted into whole number by multiplying with the suitable coefficient.
Step 5: Write empirical formula by mentioning the numbers after writing the symbols of respective elements.
35. Many a time, the reactions are carried out when the reactants are not present in the amounts as required by a balanced chemical reaction. In such situations, one reactant is in excess over the other. The reactant which is present in the lesser amount gets consumed after sometime and after that no further reaction takes place whatever be the amount of the other reactant present. Hence, the reactant which gets consumed, limits the amount of product formed and is, therefore, called the limiting reagent.
36. Mass per cent =
It is the amount of solute in grams dissolved per 100 g of solution. e.g., 10% solution of sodium chloride means 10 g of solid sodium chloride present in 100 g of solution.
37. Mole Fraction: It is ratio of number of moles of a particular component to the total number of moles of all the components.
in case of dilute solution
38. Molality (m): It is defined as number of moles of solute (B) per 1000 g or 1 kg of solve.
where is mass of solvent.
39. Molarity (M): It is expressed as the number of moles of solute per litre of solution.
where is mass of solvent.
Some Basic Concepts of Chemistry class 11 Notes
- CBSE Revision notes (PDF Download) Free
- CBSE Revision notes for Class 11 Chemistry PDF
- CBSE Revision notes Class 11 Chemistry – CBSE
- CBSE Revisions notes and Key Points Class 11 Chemistry
- Summary of the NCERT books all chapters in Chemistry class 11
- Short notes for CBSE class 11th Chemistry
- Key notes and chapter summary of Chemistry class 11
- Quick revision notes for CBSE board exams
CBSE Class-11 Revision Notes and Key Points
Some Basic Concepts of Chemistry class 11 Notes Chemistry. CBSE quick revision note for class-11 Chemistry, Physics, Maths, Biology and other subject are very helpful to revise the whole syllabus during exam days. The revision notes covers all important formulas and concepts given in the chapter. Even if you wish to have an overview of a chapter, quick revision notes are here to do if for you. These notes will certainly save your time during stressful exam days.
- Revision Notes for class-11 Physics
- Revision Notes for class-11 Chemistry
- Revision Notes for class-11 Mathematics
- Revision Notes for class-11 Biology
- Revision Notes for class-11 Accountancy
- Revision Notes for class-11 Economics
- Revision Notes for class-11 Business Studies
- Revision Notes for class-11 Computer Science
- Revision Notes for class-11 Informatics Practices
- Revision Notes for class-11 Geography
To download Some Basic Concepts of Chemistry class 11 Notes, sample paper for class 11 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
- Some Basic Concepts of Chemistry class 11 Notes Chemistry
- Structure of Atom class 11 Notes Chemistry
- Classification of Elements and Periodicity in Properties class 11 Notes Chemistry
- Chemical Bonding and Molecular Structure class 11 Notes Chemistry
- States of Matter class 11 Notes Chemistry
- Thermodynamics class 11 Notes Chemistry
- Equilibrium class 11 Notes Chemistry
- Redox Reactions class 11 Notes Chemistry
- Hydrogen class 11 Notes Chemistry
- The s-Block Elements class 11 Notes Chemistry
- The p-Block Elements class 11 Notes Chemistry
- Organic Chemistry – Some Basic Principles and Techniques class 11 Notes Chemistry
- Hydrocarbons class 11 Notes Chemistry
- Environmental Chemistry class 11 Notes Chemistry

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now