Download CBSE sample paper for class 12 Chemistry from myCBSEguide. class 12 Chemistry sample papers for board exams are available for download in myCBSEguide app, the best app for CBSE students. Sample Paper for class 12 Chemistry includes questions from Chemistrty Part-I Published by NCERT, Chemistry Part-II Published by NCERT. CBSE conducts board exam for CBSE students which will cover the whole book.
Download Complete set of sample paper for class 12 Chemistry
For study on the go download myCBSEguide app for android phones. Sample paper for class 12 Chemistry and other subjects are available for download as PDF in app too.
Chemistry Sample papers
Here is CBSE class 12 Sample paper for Chemistry. To get the answers and more sample papers, visit myCBSEguide App or website stated above. Sample paper for class 12 Chemistry has 26 questions.
Sample Paper for class 12 Chemistry
Session 2017-2018
General Instructions:
- All questions are compulsory.
- Questions number 1 to 5 are very short answer questions and carry 1 mark each.
- Questions number 6 to 10 are short answer questions and carry 2 marks each.
- Questions number 11 to 22 are also short answer questions and carry 3 marks each.
- Question number 23 is a value based question and carry 4 marks.
- Questions number 24 to 26 are long answer questions and carry 5 marks each.
- Use log tables, if necessary. Use of calculators is not allowed
1. (CH3)3C—CHO does not undergo aldol condensation. Comment.(1)
2. In the process of wine making, ripened grapes are crushed so that sugar and enzyme should come in contact with each other and fermentation should start. What will happen if anaerobic conditions are not maintained during this process?(1)
3. A coordination compound with molecular formula CrCl3.4H2O precipitates one mole of AgCl with AgNO3 solution. Its molar conductivity is found to be equivalent to two ions. What is the structural formula and name of the compound?(1)
4. How is Brownian movement responsible for the stability of sols?(1)
5. In the Arrhenius equation, what does the factor {tex}e^ {{-E}_{a}}/ {RT}{/tex} corresponds to?(1)
6. (i) Allyl cholride can be distinguished from Vinyl chloride by NaOH and silver nitrate test. Comment.(2)
(ii) Alkyl halide reacts with Lithium aluminium hydride to give alkane. Name the attacking reagent which will bring out this change.
7. Which of the following solutions has higher freezing point?
0.05 M Al2(SO4)3, 0.1 M K3[Fe(CN)6] Justify.(2)
8. Calculate the emf of the following cell at 298 K:(2)
Cr(s) / Cr3+ (0.1M) // Fe2+ (0.01M) / Fe(s)
[Given: {tex}E_{cell}^0 = + 0.30V{/tex}]
OR
The conductivity of 10-3 mol /L acetic acid at 25°C is 4.1 × 10-5 S cm-1. Calculate its degree of dissociation, if {tex} \wedge _m^0{/tex} for acetic acid at 25°C is 390.5 S cm2 mol-1.
9. What happens when:(2)
(i) Orthophosphorus acid is heated?
(ii) XeF6 undergoes complete hydrolysis?
10. Identify the following:(2)
(i) Oxoanion of chromium which is stable in acidic medium.
(ii) The lanthanoid element that exhibits +4 oxidation state.
11. Give the IUPAC name of the product formed when:(3)
(i) 2-Methyl-1-bromopropane is treated with sodium in the presence of dry ether.
(ii) 1- Methyl cyclohexene is treated with HI.
(iii) Chloroethane is treated with silver nitrite.
12. The freezing point of benzene decreases by 2.12 K when 2.5 g of benzoic acid (C6H5COOH) is dissolved in 25 g of benzene. If benzoic acid forms a dimer in benzene, calculate the van’t Hoff factor and the percentage association of benzoic acid. (Kf for benzene = 5.12 K kg mol-1):(3)
13. Explain the following behaviours::(3)
(i) Alcohols are more soluble in water than the hydrocarbons of comparable molecular masses.
(ii) Ortho-nitrophenol is more acidic than ortho-methoxyphenol.
(iii) Cumene is a better starting material for the preparation of phenol.
14. The rate constant for a first order reaction is 60 s-1. How much time will it take to reduce 1g of the reactant to 0.0625 g?:(3)
15. (i) Solutions of two electrolytes ‘A’ and ‘B’ are diluted.:(3)
The limiting molar conductivity of ‘B’ increases 1.5 times while that of ‘A’ increases 25 times. Which of the two is a strong electrolyte? Justify your answer.
(ii) The products of electrolysis of aqueous NaCl at the respective electrodes are:
Cathode : H2
Anode : Cl2 and not O2. Explain.
16. (i) Write the expression for Freundlich’s equation to describe the behaviour of adsorption from solution.:(3)
(ii) What causes charge on sol particles?
(iii) Name the promoter used in the Haber’s process for the manufacture of ammonia.
17. An organic aromatic compound ‘A’ with the molecular formula C6H7N is sparingly soluble in water. ‘A’ on treatment with dil HCI gives a water soluble compound ‘B’. ‘A’ also reacts with chloroform in presence of alcoholic KOH to form an obnoxious smelling compound ‘C’. ‘A’ reacts with benzene sulphonyl chloride to form an alkali soluble compound ‘D’.’A’ reacts with NaNO2 and HCl to form a compound ‘E’ which on reaction with phenol forms an orange red dye ‘F’. Elucidate the structures of the organic compounds from ‘A’ to ‘F’.:(3)
18. (i) Which vitamin deficiency causes rickets?:(3)
(ii) Name the base that is found in nucleotide of RNA only.
(iii) Glucose on reaction with acetic acid gives glucose penta acetate. What does it suggest about the structure of glucose?
19. Name the type of reaction involved in the formation of the following polymers from their respective monomers:(3)
(i) PVC.
(ii) Nylon6.
(iii) PHBV.
20. Describe the role of:(3)
(i) NaCN in the extraction of gold from its ore.
(ii) Cryolite in the extraction of aluminium from pure alumina.
(iii) CO in the purification of Nickel
21. A metal ion Mn+ having d4 valence electronic configuration combines with three bidentate ligands to form a complex compound. Assuming ∆a > P:(3)
(i) Write the electronic configuration of d4 ion.
(ii) What type of hybridisation will Mn+ ion has?
(iii) Name the type of isomerism exhibited by this complex.
22. The magnetic moments of few transition metal ions are given below::(3)
|
Metal ion |
Magnetic moment (BM) |
|
SC3+ |
0.00 |
|
Cr2+ |
4.90 |
|
Ni2+ |
2.84 |
|
Ti3+ |
1.73 |
(at no. Sc = 21, Ti =22, Cr = 24, Ni = 28)
Which of the given metal ions :
(i) has the maximum number of unpaired electrons?
(ii) forms colourless aqueous solution?
(iii) exhibits the most stable +3 oxidation state?
OR
Consider the standard electrode potential values (M2+ / M) of the elements of the first transition series.
| Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
| -1.63 | -1.18 | -0.90 | -1.18 | -0.44 | -0.28 | -0.25 | +0.34 | -0.76 |
Explain:
(i) E° value for copper is positive.
(ii) E° value of Mn is more negative as expected from the trend.
(iii) Cr2+ is a stronger reducing agent than Fe2+.
23. Ashwin observed that his friend Shubham was staying aloof, not playing with friends and becoming easily irritable for some weeks. Ashwin told his teacher about this, who, in turn, called Shubham’s parents and advised them to consult a doctor. Doctor after examining Shubham prescribed antidepressant drugs for him.
After reading the above passage, answer the following questions:(4)
i) Name two antidepressant drugs.
ii) Mention the values shown by Ashwin.
iii) How should Shubham’s family help him other than providing medicine?
iv) What is the scientific explanation for the feeling of depression?
24. (a) Arrange the following in the order of property indicated against each set:(5)
(i) F2, Cl2, Br2, I2 (increasing bond dissociation enthalpy)
(ii) H2O, H2S, H2Se, H2Te (increasing acidic character)
(b) A colourless gas ‘A’ with a pungent odour is highly soluble in water and its aqueous solution is weakly basic. As a weak base it precipitates the hydroxides of many metals from their salt solution. Gas ‘A’ finds application in detection of metal ions. It gives a deep blue colouration with copper ions. Identify the gas ‘A’ and write the chemical equations involved in the following:
(i) Gas ‘A’ with copper ions
(ii) Solution of gas ‘A’ with ZnSO4 solution.
OR
Answer the following questions
(a) Write the formula of the neutral molecule which is isoelectronic with ClO–.
(b) Draw the shape of H2S2O7.
(c) Nitric acid forms an oxide of nitrogen on reaction with P4. Write the formula of the stable molecule formed when this oxide undergoes dimerisation.
(d) Bleaching action of chlorine is permanent. Justify.
(e) Write the disproportionation reaction of that oxoacid of nitrogen in which nitrogen is in +3 oxidation state.
25. Write the products of the following reactions:(5)
(i) 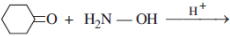
(ii) {tex}2C_6{H_5}CHO + conc.NaOH \to {/tex}
(iii) {tex}C{H_3}COOH\xrightarrow{{C{l_2}/P}}{/tex}
(b) Give simple chemical tests to distinguish between the following pairs of compounds:
(i) Benzaldehyde and Benzoic acid
(ii) Propanal and Propanone
OR
(a) Account for the following:
(i) CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
(ii) 2-Fluorobutanoic acid is a stronger acid than 3-Fluorobutanoic acid.
(b) Write the chemical equations to illustrate the following name reactions:
(i) Etard reaction.
(ii) Rosenmund’s reaction.
(c) Give the mechanism of cyanohydrin formation when carbonyl compounds react with HCN in the presence of alkali.
26. (i) Following is the schematic alignment of magnetic moments:(5)

Identify the type of magnetism. What happens when these substances are heated?
(ii) If the radius of the octahedral void is ‘r’ and radius of the atoms in close packing is ‘R’. What is the relation between ‘r’ and ‘R’?
(iii) Tungsten crystallizes in body centred cubic unit cell. If the edge of the unit cell is 316.5 pm. What is the radius of tungsten atom?
OR
(i) Identify the type of defect shown in the following figure:

What type of substances show this defect?
(ii) A metal crystallizes in a body centred cubic structure. If ‘a’ is the edge length of its unit cell, ‘r’ is the radius of the sphere. What is the relationship between ‘r’ and ‘a’?
(iii) An element with molar mass 63 g / mol forms a cubic unit cell with edge length of 360.8 pm. If its density is 8.92 g/ cm3. What is the nature of the cubic unit cell?
This is only initial part of the whole sample paper. Download Complete set of sample paper for class 12 Chemistry
Sample Paper for class 12
It is sample paper for class 12 Chemistry. However, myCBSEguide provides the best sample papers for all the subjects. There are number of sample papers which you can download from myCBSEguide website. Sample paper for class 12 all subjects
- CBSE Sample Papers for Class 12 Physics
- CBSE Sample Papers for Class 12 Chemistry
- CBSE Sample Papers for Class 12 Mathematics
- CBSE Sample Papers for Class 12 Biology
- CBSE Sample Papers for Class 12 English Core
- CBSE Sample Papers for Class 12 Business Studies
- CBSE Sample Papers for Class 12 Economics
- CBSE Sample Papers for Class 12 Accountancy
- CBSE Sample Papers for Class 12 Computer Science
- CBSE Sample Papers for Class 12 Informatics Practices
- CBSE Sample Papers for Class 12 Hindi Core
- CBSE Sample Papers for Class 12 Hindi Elective
- CBSE Sample Papers for Class 12 History
- CBSE Sample Papers for Class 12 Political Science
- CBSE Sample Papers for Class 12 Geography
- CBSE Sample Papers for Class 12 Home Science
- CBSE Sample Papers for Class 12 Physical Education
- CBSE Sample Papers for Class 12 Other Subjects
These are also Sample Paper for class 12 Chemistry available for download through myCBSEguide app. These are the latest Sample Paper for class 12 Chemistry as per the new board exam pattern. Download the app today to get the latest and up-to-date study material.
Marking Scheme for Class 12 Board exam
| Subject | Board Marks | Practical or internal Marks |
| English | 100 Marks | ZERO Marks |
| Hindi | 100 Marks | ZERO Marks |
| Mathematics | 100 Marks | ZERO Marks |
| Chemistry | 70 Marks | 30 Marks |
| Physics | 70 Marks | 30 Marks |
| Biology | 70 Marks | 30 Marks |
| Computer Science | 70 Marks | 30 Marks |
| Informatics Practices | 70 Marks | 30 Marks |
| Accountancy | 80 Marks | 20 Marks |
| Business Studies | 80 Marks | 30 Marks |
| Economics | 80 Marks | 20 Marks |
| History | 80 Marks | 20 Marks |
| Political Science | 100 Marks | ZERO Marks |
| Geography | 70 Marks | 30 Marks |
| Sociology | 80 Marks | 20 Marks |
| Physical Education | 70 Marks | 30 Marks |
| Home Science | 70 Marks | 30 Marks |
Download CBSE Sample Papers
To download complete sample paper for class 12 Chemistry, Physics, Economics, Mathematics, Biology, English Core, Business Studies, Accountancy, Computer Science and Political Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now