NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2 Class 9 Science book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 9 Science chapter wise NCERT solution for Science Book for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Science Is Matter around Us Pure Download as PDF

NCERT Class 9 Science Chapter wise Solutions
- 01 – Matter in Our Surroundings
- 02 – Is Mattter Around us Pure
- 03 – Atoms and Molecules
- 04 – Structure of the Atom
- 05 – The Fundamental Unit of Life
- 06 – Tissues
- 07 – Diversity in Living Organisms
- 08 – Motion
- 09 – Force and Laws of Motion
- 10 – Gravitation
- 11 – Word and Energy
- 12 – Sound
- 13 – Why Do We Fall Ill
- 14 – Natural Resources
- 15 – Improvement in Food Resources
NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
1. Which separation techniques will you apply for the separation of the following?
(a) Sodium chloride from its solution in water.
(b) Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flower petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Ans. (a) Evaporation method
(b) Sublimation method
(c) by heating and then after filtration
(d) by Chromatography
(e)by method of centrifugation
(f) by using separating funnel
(g) by filtration method using strainer
(h) with the help of a magnet
(i) by winnowing
(j) by centrifugation
2. Write the steps you would use for making tea. Use the words solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
Ans. Take more amount of solvent (water) in a pan and after heating it add little amount of solute (sugar) to the solvent. Solute will dissolve completely in the solvent forming true solution, then add tea leaves that are insoluble along with another soluble liquid milk. After boiling allow filtration with a sieve so the filtrate you obtain is tea while the residue has tea leaves that are thrown away.
NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
3. Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution).
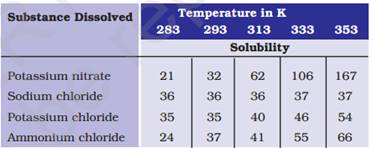
(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
(b) Pragya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
(c) Find the solubility of each salt at 293 K. Which salt has the highest solubility at this temperature?
(d) What is the effect of change of temperature on the solubility of a salt?
Ans. (a) At 313 K temperature the amount of potassium nitrate required was 62g in 100ml of water so in 50g water we will need to dissolve = 62 X 50/100= 31g potassium nitrate.
(b) At 373K saturated solution preparation needs 54g potassium nitrate and at room temperature (293 K) saturation solution formation occurs with 35g potassium nitrate hence = 54 – 35 = 19g potassium nitrate will precipitate out as undissolved salt.
(c) Solubilities are (in 100mg of water) 32,36,35,37 respectively for the mentioned salts and the highest solubility is of ammonium chloride at this temperature.
(d) Solubility of salts is directly proportional to the temperature i.e. if temperature increases then solubility will increase and if the temperature decreases solubility will also decrease.
NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
4. Explain the following giving examples.
(a) saturated solution
(b) pure substance
(c) colloid
(d) suspension
Ans. (a) saturated solution :- It is a solution in which no more solute particles can be dissolved at a particular temperature.
(b) pure substance :- Such substance that has a uniform composition i.e. has particles with identical properties is called pure substance eg sugar, salt, water, nitrogen etc.
(c) colloid :- It is a kind of heterogeneous mixture/solution in which particle size is between 1nm and 1000nm. Colloids have dispersion medium and dispersed phase.eg smoke, milk, shaving cream, jelly, cheese etc.
(d) suspension :- It is a kind of heterogeneous mixture in which insoluble solid particles remain suspended in the medium and dispersion particles are visible to the unaided eyes. eg muddy river water, chalk powder in water, dust storm, sand in water etc.
5. Classify each of the following as a homogeneous or heterogeneous mixture.
soda water, wood, air, soil, vinegar, filtered tea.
Ans.

NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
6. How would you confirm that a colourless liquid given to you is pure water?
Ans. If we allow the given liquid to evaporate by heating it as in a clean china dish so:
O any residue remaining in the china dish will indicate that water is not pure but contains impurities.
O no residue in china dish will indicate that water is pure.
7. Which of the following materials fall in the category of a “pure substance”?
(a) Ice
(b) Milk
(c)Iron
(d)Hydrochloric acid
(e) Calcium oxide
(f) Mercury
(g) Brick
(h) Wood
(i) Air.
Ans. Pure substances are: ice, iron, hydrochloric acid, calcium oxide, mercury.
8. Identify the solutions among the following mixtures.
(a) Soil
(b) Sea water
(c) Air
(d) Coal
(e) Soda water.
Ans. Sea water, air and soda water are solutions.
9. Which of the following will show “Tyndall effect”?
(a) Salt solution
(b)Milk
(c) Copper sulphate solution
(d) Starch solution.
Ans. Milk and starch solution have larger particles since they are not true solutions so they will show tyndall effect.
NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
10. Classify the following into elements, compounds and mixtures.
(a) Sodium
(b) Soil
(c) Sugar solution
(d) Silver
(e) Calcium carbonate
(f) Tin
(g) Silicon
(h) Coal
(i) Air
(j) Soap
(k) Methane
(l) Carbon dioxide
(m) Blood
Ans.

NCERT Solutions for Class 9 Science Is Matter around Us Pure part 2
11. Which of the following are chemical changes?
(a) Growth of a plant
(b) Rusting of iron
(c) Mixing of iron filings and sand
(d) Cooking of food
(e) Digestion of food
(f) Freezing of water
(g) Burning of a candle.
Ans. Rusting of iron, cooking of food, digestion of food, burning of a candle are chemical changes.
NCERT Solutions for Class 9 Science
NCERT Solutions Class 9 Science PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 9 Science includes text book solutions .NCERT Solutions for CBSE Class 9 Science have total 15 chapters. 9 Science NCERT Solutions in PDF for free Download on our website. Ncert Science class 9 solutions PDF and Science ncert class 9 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide.
CBSE app for Class 9
To download NCERT Solutions for class 9 Science, Computer Science, Home Science,Hindi ,English, Maths Social Science do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now