NCERT Solutions for Class 9 Science Atoms and Molecules part 2 Class 9 Science book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 9 Science chapter wise NCERT solution for Science Book for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Science Atoms and Molecules Download as PDF

NCERT Class 9 Science Chapter wise Solutions
- 01 – Matter in Our Surroundings
- 02 – Is Mattter Around us Pure
- 03 – Atoms and Molecules
- 04 – Structure of the Atom
- 05 – The Fundamental Unit of Life
- 06 – Tissues
- 07 – Diversity in Living Organisms
- 08 – Motion
- 09 – Force and Laws of Motion
- 10 – Gravitation
- 11 – Word and Energy
- 12 – Sound
- 13 – Why Do We Fall Ill
- 14 – Natural Resources
- 15 – Improvement in Food Resources
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
1.A 0.24 g sample of compound of oxygen and boron was found by analysis to contain 0.096 g of boron and 0.144 g of oxygen. Calculate the percentage composition of the compound by weight.
Ans. Mass of the given sample compound = 0.24g
Mass of boron in the given sample compound = 0.096g
Mass of oxygen in the given sample compound = 0.144g
% composition of compound = % of boron and % of oxygen
Therefore % of boron = mass of boron 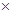 100/mass of the sample compound
100/mass of the sample compound
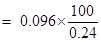
= 40%
Therefore % of oxygen = mass of oxygen 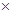 100/mass of the sample compound
100/mass of the sample compound
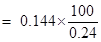
= 60%
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
2.When 3.0 g of carbon is burnt in 8.00 g oxygen, 11.00 g of carbon dioxide is produced. What mass of carbon dioxide will be formed when 3.00 g of carbon is burnt in 50.00 g of oxygen? Which law of chemical combination will govern your answer?
Ans. According to the law of chemical combination of constant proportions “in a chemical compound the elementary constituents always combine in constant proportions by weight/mass”. Therefore, whether 3 g carbon is burnt in 8 g oxygen or 3g carbon is burnt in 50g oxygen in both cases only 11g carbon dioxide will be formed.
3.What are polyatomic ions? Give examples.
Ans. When two or more atoms in a group is having a charge, such is called a polyatomic ion. For 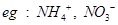 etc.
etc.
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
4.Write the chemical formulae of the following.
(a) Magnesium chloride
(b) Calcium oxide
(c) Copper nitrate
(d) Aluminium chloride
(e) Calcium carbonate.
Ans.

5.Give the names of the elements present in the following compounds.
(a) Quick lime
(b) Hydrogen bromide
(c) Baking powder
(d) Potassium sulphate.
Ans.

NCERT Solutions for Class 9 Science Atoms and Molecules part 2
6. Calculate the molar mass of the following substances.
(a) Ethyne, 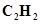
(b) Sulphur molecule, 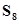
(c) Phosphorus molecule, 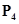 (Atomic mass of phosphorus= 31)
(Atomic mass of phosphorus= 31)
(d) Hydrochloric acid, HCl
(e) Nitric acid, 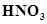
Ans. (a)Ethyene = 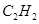
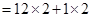 =24 + 2= 26 u =26 g
=24 + 2= 26 u =26 g
(b)Sulphur molecular = 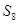
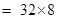 = 256 u= 256 g
= 256 u= 256 g
(c)Phosphorus molecule = 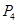
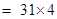 = 124 u= 124 g
= 124 u= 124 g
(d)Hydrochloric acid = HCl = 1+ 35.5 = 36.5 u= 36.5 g
(e)Nitric acid = 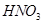 = 1 + 14 +
= 1 + 14 + 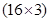 = 15 + 48 = 63 u =63 g
= 15 + 48 = 63 u =63 g
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
7.What is the mass of—
(a) 1 mole of nitrogen atoms?
(b) 4 moles of aluminium atoms (Atomic mass of aluminium= 27)?
(c) 10 moles of sodium sulphite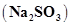 ?
?
Ans. (a)Atomic mass of nitrogen is 14 u.
therefore 1 mol of N = 14g
(b)Atomic mass of aluminium = 27u
therefore 1 mol of Al = 27g and so 4 mol of Al 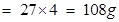
(c)molecular mass of 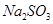
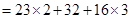 = 46 + 32 + 48 = 126 u
= 46 + 32 + 48 = 126 u
therefore 1 mol of 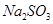 has weight/mass 126g.
has weight/mass 126g.
hence, 10 mol of 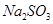
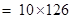 = 1260g
= 1260g
8.Convert into mole.
(a) 12 g of oxygen gas
(b) 20 g of water
(c) 22 g of carbon dioxide.
Ans. (a)molecular mass of 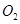 = 32 u= 32g(1 mole)
= 32 u= 32g(1 mole)
since 32 g of 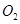 =1mole then 12g of
=1mole then 12g of 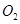
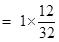 =0.375mole.
=0.375mole.
(b)molecular mass of 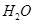
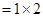 + 16 = 18 u= 18g(1mole)
+ 16 = 18 u= 18g(1mole)
20g 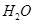
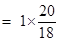 = 1.11mole.
= 1.11mole.
(c)molecular mass of 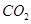
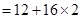 = 12 + 32 = 44 u= 44g (1mole)
= 12 + 32 = 44 u= 44g (1mole)
22g of 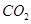
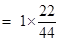 = 0.5mole.
= 0.5mole.
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
9.What is the mass of:
(a) 0.2 mole of oxygen atoms?
(b) 0.5 mole of water molecules?
Ans. (a) since 1 mole of O = atomic mass of O = 16u=16g
then 0.2mole of 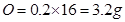
(b)1mol of H2O = molecular mass of 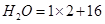 =18 u =18g
=18 u =18g
then 0.5mol of 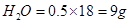
10.Calculate the number of molecules of sulphur (S8) present in 16 g of solid sulphur.
Ans. 1mol of 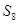 = molecular mass of
= molecular mass of 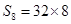 = 256u=256g
= 256u=256g
since 256g of 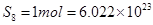 atoms (Avogadro number)
atoms (Avogadro number)
16g of 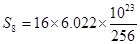
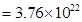 molecules
molecules
NCERT Solutions for Class 9 Science Atoms and Molecules part 2
11.Calculate the number of aluminium ions present in 0.051 g of aluminium oxide.
(Hint: The mass of an ion is the same as that of an atom of the same element. Atomic mass of Al = 27 u)
Ans. 1mol of 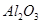 = molecular mass of
= molecular mass of 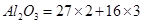 = 102u=102g
= 102u=102g
aluminium ions present in 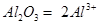
102 g of 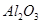 contains aluminium ions
contains aluminium ions 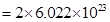
then 0.051 g 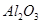 contains aluminium ions
contains aluminium ions 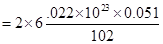
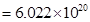
NCERT Solutions for Class 9 Science
NCERT Solutions Class 9 Science PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 9 Science includes text book solutions .NCERT Solutions for CBSE Class 9 Science have total 15 chapters. 9 Science NCERT Solutions in PDF for free Download on our website. Ncert Science class 9 solutions PDF and Science ncert class 9 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide.
CBSE app for Class 9
To download NCERT Solutions for class 9 Science, Computer Science, Home Science,Hindi ,English, Maths Social Science do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now