NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2 Class 10 Science Class book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE Board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 10 Science chapter wise NCERT solution for Science Book for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Class 10 Science Periodic Classification of Elements Download as PDF

NCERT Class 10 Science Chapter wise Solutions
- 1 – Chemical Reactions and Equations
- 2 – Acids, Bases and Salts
- 3 – Metals and Non-metals
- 4 – Carbon and Its Compounds
- 5 – Periodic Classification of Elements
- 6 – Life Processes
- 7 – Control and Coordination
- 8 – How do Organisms Reproduce?
- 9 – Heredity and Evolution
- 10 – Light Reflection and Refraction
- 11 – Human Eye and Colourful World
- 12 – Electricity
- 13 – Magnetic Effects of Electric Current
- 14 – Sources of Energy
- 15 – Our Environment
- 16 – Management of Natural Resources
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
1. Which of the following statements is not correct statement about the trends when going from left to right across the periods of Periodic Table?
(a) The elements become less metallic in nature.
(b) The number of valance electrons increases.
(c) The atoms lose their electrons more easily.
(d) The oxides becomes more acidic.
Ans. (c) The atoms lose their electrons more easily.
2. Element X forms a chloride with the formula 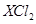 which is a solid with a high melting point. X would most likely be in the same group of the Periodic Table as
which is a solid with a high melting point. X would most likely be in the same group of the Periodic Table as
(a) Na
(b) Mg
(c) Al
(d) Si
Ans. (b) Mg
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
3. Which element has
(a) two cells, both of which are completely filled with electrons?
(b) the electronic configuration 2, 8, 2?
(c) a total of three shells, with four electrons in its valance shell?
(d) twice as many electrons in its second shell as in its first shell?
Ans. (a) Neon
(b) Magnesium
(c) Silicon
(d) Carbon
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
4. (a) What property do all elements in the same column of the Periodic Table as boron have in common?
(b) What property do all elements in the same column of the Periodic Table as Fluorine have common?
Ans. (a) All the elements in the in the same column as boron have three electrons in the valence shell. That is all the elements are trivalent.
(b) All the elements in the same column as fluorine has one electron in the valance shell, which is all the elements, are monovalent.
5. An atom has electronic configuration 2, 8, 7.
(a) What is the atomic number of this element?
(b)To which of the following elements would it be chemically similar?
(Atomic numbers are given in parentheses.)
N(7), F(9), P(15), Ar(18)
Ans. Chlorine has the electronic configuration 2, 8, 7.
(a) Atomic number of element is 17.
(b) F (9)
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
6. The position of three elements A, B and C in the Periodic Table are shown below:

(a) State whether A is a metal or non-metal.
(b) State whether C is more reactive than A
(c) Will C be larger or smaller in size than B?
(d) Which type of ion, cation or anion, will be formed by element A?
Ans. (a) A is a non-metal.
(b) C is less reactive than A.
(c) C will be smaller than B.
(d) A will form anion.
7. Nitrogen (atomic number 7) and phosphorus (atomic number 15) belong to group 15 of the Periodic Table. Write the electronic configuration of these two elements. Which of these will be more electronegative? Why?
Ans. Nitrogen atomic number 7 has got 7 electrons with electronic configuration 2, 5.
Phosphorus with atomic number 15 has got 15 electrons with electronic configuration 2, 8, 5.
Non-metallic character decreases as we move down the group. Therefore, nitrogen will be more electronegative than phosphorus.
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
8. How does the electronic configuration of an atom relate to its position in the Modern Periodic Table?
Ans. Group number on an element can be predicted from the number of electrons in the outermost shell.
Period number of an element can be predicted from the number of shells with filled electrons.
Knowing the electronic configuration, we can find the number of electrons in the outermost shell and the number of shells with filled electrons. This can help to relate its position in the Periodic Table.
9. In the modern Periodic Table calcium (atomic number 20) is surrounded by elements with atomic number 12, 19, 21 and 38. Which of these have physical and chemical properties resembling calcium?
Ans. Elements in a group have similar properties. Elements with atomic numbers 12 and 38 lie in the same group as calcium. Therefore, they will have properties resembling calcium.
NCERT Solutions for Class 10 Science Periodic Classification of Elements Part 2
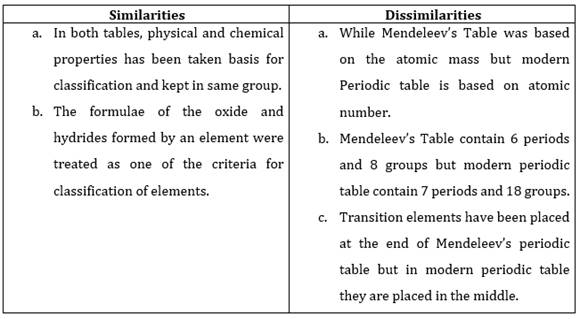
10. Compare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table.
Ans.
NCERT Solutions for Class 10 Science
NCERT Solutions Class 10 Science PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 10 Science includes text book solutions from Book. NCERT Solutions for CBSE Class 10 Science have total 16 chapters. 10 Science NCERT Solutions in PDF for free Download on our website. Ncert Science class 10 solutions PDF and Science ncert class 10 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide.
CBSE app for Students
To download NCERT Solutions for class 10 Social Science, Computer Science, Home Science,Hindi ,English, Maths Science do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now