NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2 Class 10 Science Class book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE Board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 10 Science chapter wise NCERT solution for Science Book for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
NCERT solutions for Class 10 Science Carbon and Its Compounds Download as PDF

NCERT Class 10 Science Chapter wise Solutions
- 1 – Chemical Reactions and Equations
- 2 – Acids, Bases and Salts
- 3 – Metals and Non-metals
- 4 – Carbon and Its Compounds
- 5 – Periodic Classification of Elements
- 6 – Life Processes
- 7 – Control and Coordination
- 8 – How do Organisms Reproduce?
- 9 – Heredity and Evolution
- 10 – Light Reflection and Refraction
- 11 – Human Eye and Colourful World
- 12 – Electricity
- 13 – Magnetic Effects of Electric Current
- 14 – Sources of Energy
- 15 – Our Environment
- 16 – Management of Natural Resources
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
1. Ethane, with the molecular formula C2H6 has
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Ans. (b) 7 covalent bonds
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
2. Butanone is a four carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol
Ans. (c) Ketone
3. While cooking, if the bottom of the vessels is getting blackened on the outside, it means that
(a) the fuel is not cooked completely.
(b) the fuel is not burning completely.
(c) the fuel is wet.
(d) the is burning completely.
Ans. (b) the fuel is not burning completely.
4. Explain the nature of the covalent bond using the bond formation in CH3Cl.
Ans. Covalent bond is formed by sharing of electrons between two atoms. It is non-ionic in nature.
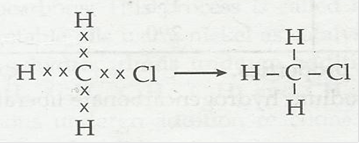
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
5. Draw the electron dot structure for
(a) Ethanoic acid
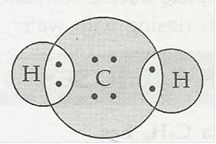
(b) H2S
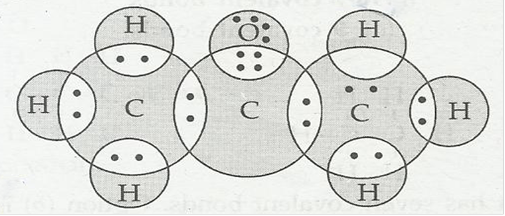
(c) Propanone
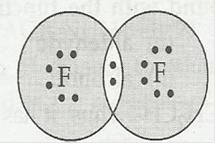
(d) F2
Ans. (a) Ethanoic acid
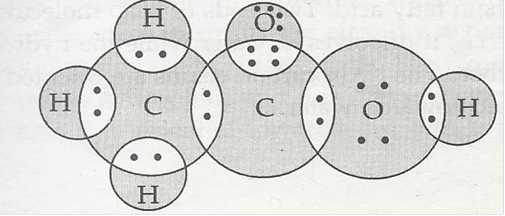
(b) H2S
(c) Propanone
(d) F2
6. What is a homologous series? Explain with an example.
Ans. Series of compounds in which the same functional group substitutes for hydrogen in a carbon chain is called homologues series. The difference between the formulae of any two successive members is –CH2 and difference between the molecular formula is 14 u.
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
7. How can ethanol and Ethanoic acid be differentiated on the basis of their physical and chemical properties?
Ans. On the basis of physical properties:
Melting and boiling points of ethanol is 156 K and 351 K but melting and boiling point of Ethanoic acid is 290K and 391K respectively.
On the chemical properties:
Ethanoic acid reacts with sodium hydrogen carbonate liberating carbon dioxide while ethanol does not.
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Ans. Soap is sodium or potassium salt of long chain fatty acid. Two ends of soap molecules have different properties. The ionic end is hydrophilic. It dissolve in water while the hydrogen chain is hydrophobic, it dissolve in hydrocarbon. The hydrocarbon chains are oriented towards the oil droplet while the ionic ends are oriented towards water.
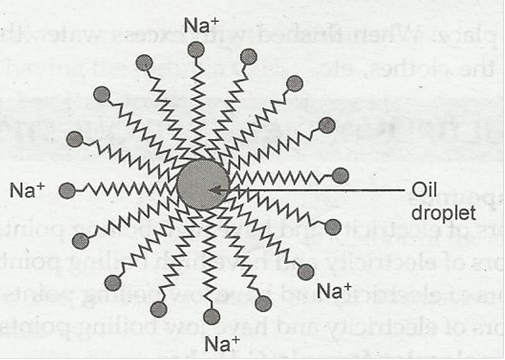
Micelles formation will not take place in ethanol.
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
9. Why are carbon and its compounds used as fuels for most applications?
Ans. Carbon on combustion gives carbon dioxide and water. This reaction is accompanied by evolution of heat and light. The same is true for compounds of carbon. That is why carbon and its compounds are used as fuel for most applications.
10. Explain the formation of scum when hard water is treated with soap.
Ans. Hard water contains hydrogen carbonates, chlorides and sulphates of calcium and magnesium which reacts with soap to form scum. For example, calcium chloride reacts with soap to form scum.
Sodium stearate + Calcium chloride à sodium chloride + Calcium stearate(scum)
11. What change will you observe if you test soap with litmus paper (red and blue)?
Ans. Soap is sodium or potassium salt of fatty acid. It is obtained by treating of oil with caustic soda. Sodium stearate is thus a salt of weak acid and strong base. Its water solution will be slightly alkaline and will turn red litmus red.
12. What is hydrogenation? What is its industrial application?
Ans. Unsaturated hydrocarbons add hydrogen in presence of catalysts such as palladium or nickel to give saturated hydrocarbons. This process is called hydrogenation.
It is commercially used for converting vegetable oils to ‘vanaspati’ ghee in presence of nickel as catalyst.
13. Which of the following hydrocarbons undergo addition reactions?
C2H6, C3H8, C3H6, C2H2 and CH4
Ans. C3H6 and C2H2 will undergo addition reactions.
14. Give a test that can be used to differentiate chemically between butter and cooking oil?
Ans. Butter and cooking oil can be differentiated with the help of bromine water test. Cooking oil will decolorize the red colour of bromine water on shaking while butter will not.
NCERT Solutions for Class 10 Science Carbon and Its Compounds Part 2
15. Explain in mechanism of the cleaning action of soap.
Ans. Soap are sodium or potassium salt of fatty acids. Two ends of molecules of soap behave differently. This ionic end is hydrophilic and it is oriented towards water. The other hydrocarbon end is hydrophobic and it is oriented towards dirt which is oily in nature. A micelle formation around the oily dirt takes place. When flushed with excess of water, the micelle containing the dirt is removed, thus cleaning the clothes, etc.
NCERT Solutions for Class 10 Science
NCERT Solutions Class 10 Science PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 10 Science includes text book solutions from Book. NCERT Solutions for CBSE Class 10 Science have total 16 chapters. 10 Science NCERT Solutions in PDF for free Download on our website. Ncert Science class 10 solutions PDF and Science ncert class 10 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide.
CBSE app for Students
To download NCERT Solutions for class 10 Social Science, Computer Science, Home Science,Hindi ,English, Maths Science do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now