NCERT Solutions class 12 Chemistry Electrochemistry part 1 solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for Electrochemistry part 1 as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d-and f-Blocks Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday Life
CHAPTER THREE ELECTROCHEMISTRY
- 3.1 Electrochemical Cells
- 3.2 Galvanic Cells
- 3.3 Nernst Equation
- 3.4 Conductance of Electrolytic Solutions
- 3.5 Electrolytic Cells and Electrolysis
- 3.6 Batteries
- 3.7 Fuel Cells
- 3.8 Corrosion
NCERT Solutions class 12 Chemistry ElectroChemistry part 1
1. Arrange the following metals in the order in which they displace each other from the solution of their salts. Al, Cu, Fe, Mg and Zn
2. Given the standard electrode potentials,
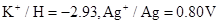 ,
,
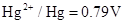
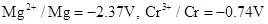
3. Depict the galvanic cell in which the reaction takes place. Further show:
(i) Which of the electrode is negatively charged?
(ii) The carriers of the current in the cell.
(iii) Individual reaction at each electrode.
4. Calculate the standard cell potentials of galvanic cells in which the following reactions take place:
(i) 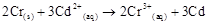
(ii) 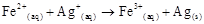
Calculate the 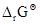 ¸ and equilibrium constant of the reactions.
¸ and equilibrium constant of the reactions.
5. Write the Nernst equation and emf of the following cells at 298 K:
(i) 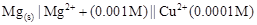
(ii) 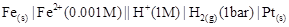
(iii) 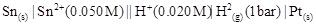
(iv) 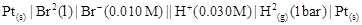
6. In the button cells widely used in watches and other devices the following reaction takes place: 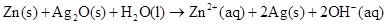 Determine
Determine 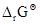 and
and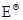 for the reaction.
for the reaction.
7. Define conductivity and molar conductivity for the solution of an electrolyte. Discuss their variation with concentration.
8. The conductivity of 0.20 M solution of KCl at 298 K is 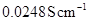 . Calculate its molar conductivity.
. Calculate its molar conductivity.
9. The resistance of a conductivity cell containing 0.001M KCl solution at 298 K is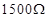 . What is the cell constant if conductivity of 0.001M KCl solution at 298 K is
. What is the cell constant if conductivity of 0.001M KCl solution at 298 K is 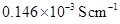 .
.
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes textbook solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide NCERT Solutions class 12 Chemistry Amines part 1
CBSE app for Class 12
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now