
myCBSEguide App
Download the app to get CBSE Sample Papers 2023-24, NCERT Solutions (Revised), Most Important Questions, Previous Year Question Bank, Mock Tests, and Detailed Notes.
Install NowNCERT Solutions class 12 Chemistry alcohol phenol and ether part 2 Class 12 Chemistry book solutions are available in PDF format for free download. These ncert book chapter wise questions and answers are very helpful for CBSE board exam. CBSE recommends NCERT books and most of the questions in CBSE exam are asked from NCERT text books. Class 12 Chemistry chapter wise NCERT solution for Chemistry part 1 and Chemistry part 2 for all the chapters can be downloaded from our website and myCBSEguide mobile app for free.
Download NCERT solutions for alcohol phenol and ether part 2 as PDF.

NCERT Class 12 Chemistry Chapter-wise Solutions
- 1 – The Solid State
- 2 – Solutions
- 3 – Electrochemistry
- 4 – Chemical Kinetics
- 5 – Surface Chemistry
- 6 – General Principles and Processes of Isolation of Elements
- 7 – The p-Block Elements
- 8 – The d-and f-Blocks Elements
- 9 – Coordination Compounds
- 10 – Haloalkanes and Haloarenes
- 11 – Alcohols, Phenols and Ethers
- 12 – Aldehydes, Ketones and Carboxylic Acids
- 13 – Amines
- 14 – Biomolecules
- 15 – Polymers
- 16 – Chemistry in Everyday Life
CHAPTER ELEVEN ALCOHOLS, PHENOLS AND ETHERS
- 11.1 Classification
- 11.2 Nomenclature
- 11.3 Structures of Functional Groups
- 11.4 Alcohols and Phenols
- 11.5 Some Commerically Important Alcohols
- 11.6 Ethers
NCERT Solutions class 12 Chemistry alcohol phenol and ether part 2
17. Give equations of the following reactions:
(i) Oxidation of propan-1-ol with alkaline 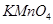 solution.
solution.
(ii) Bromine in 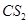 with phenol.
with phenol.
(iii) Dilute 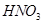 with phenol.
with phenol.
(iv) Treating phenol with chloroform in presence of aqueous NaOH.
18. Explain the following with an example.
(i) Kolbe’s reaction.
(ii) Reimer-Tiemann reaction.
(iii) Williamson ether synthesis.
(iv) Unsymmetrical ether.
19. Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
20. How are the following conversions carried out?
(i) Propene → Propan-2-ol
(ii) Benzyl chloride → Benzyl alcohol
(iii) Ethyl magnesium chloride → Propan-1-ol.
(iv) Methyl magnesium bromide → 2-Methylpropan-2-ol.
21. Name the reagents used in the following reactions:
(i) Oxidation of a primary alcohol to carboxylic acid.
(ii) Oxidation of a primary alcohol to aldehyde.
(iii) Bromination of phenol to 2,4,6-tribromophenol.
(iv) Benzyl alcohol to benzoic acid.
(v) Dehydration of propan-2-ol to propene.
(vi) Butan-2-one to butan-2-ol.
22. Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
23. Give IUPAC names of the following ethers:
(i)
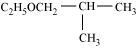
(ii)
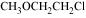
(iii)
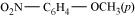
(iv)
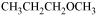
(v)
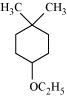
(vi)
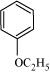
24. Write the names of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
(i) 1-Propoxypropane
(ii) Ethoxybenzene
(iii) 2-Methoxy-2-methylpropane
(iv) 1-Methoxyethane
25. Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
26. How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
27. Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
28. Write the equation of the reaction of hydrogen iodide with:
(i) 1-propoxypropane
(ii) Methoxybenzene and
(iii) Benzyl ethyl ether
29. Explain the fact that in aryl alkyl ethers
(i) The alkoxy group activates the benzene ring towards electrophilic substitution and
(ii) It directs the incoming substituents to ortho and para positions in benzene ring.
30. Write the mechanism of the reaction of HI with methoxymethane.
31. Write equations of the following reactions:
(i) Friedel-Crafts reaction-alkylation of anisole.
(ii) Nitration of anisole.
(iii) Bromination of anisole in ethanoic acid medium.
(iv) Friedel-Craft’s acetylation of anisole.
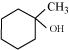
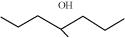
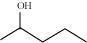
32. Show how would you synthesise the following alcohols from appropriate alkenes?
(i)
(ii)
(iii)
(iv)
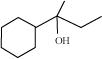
33. When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
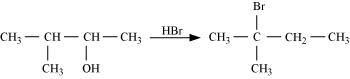
Give a mechanism for this reaction. (Hint: The secondary carbocation formed in step II rearranges to a more stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.
NCERT Solutions class 12 Chemistry
NCERT Solutions Class 12 Chemistry PDF (Download) Free from myCBSEguide app and myCBSEguide website. Ncert solution class 12 Chemistry includes text book solutions from both part 1 and part 2. NCERT Solutions for CBSE Class 12 Chemistry have total 16 chapters. 12 Chemistry NCERT Solutions in PDF for free Download on our website. Ncert Chemistry class 12 solutions PDF and Chemistry ncert class 12 PDF solutions with latest modifications and as per the latest CBSE syllabus are only available in myCBSEguide
CBSE app for Students
To download NCERT Solutions for class 12 Chemistry, Physics, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
myCBSEguide
Question Bank, Mock Tests, Exam Papers, NCERT Solutions, Sample Papers, Notes
Install Now