Chemistry in Everyday Life Class 12 Revision Notes -Free PDF Download
CBSE Class 12 Chemistry notes Chapter 16 in Everyday Life in PDF are available for free download in myCBSEguide mobile app. The best app for CBSE students now provides Chemistry in Everyday Life class 12 Notes Chemistry latest chapter wise notes for quick preparation of CBSE board exams and school-based annual examinations. Class 12 Chemistry notes on chapter 16 Chemistry in Everyday Life are also available for download in CBSE Guide website.
Chemistry in Everyday Life class 12 Notes Chemistry
Download revision notes for Chemistry in Everyday Life class 12 Notes and score high in exams. These are the class 12 Notes prepared by team of expert teachers. The revision notes help you revise the whole chapter 16 in minutes. Revision notes in exam days are one of the best tips recommended by teachers during exam days.
CBSE Class 12 Chemistry Revision Notes
Class 12 Chemistry Revision Notes Chapter 16 Chemistry in Everyday Life
- Drugs: Drugs are low molecular mass substances which interact with targets in the body and produce a biological response.
- Medicines: Medicines are chemicals that are useful in the diagnosis, prevention, and treatment of diseases
- Therapeutic effect: Desirable or beneficial effect of a drug like the treatment of symptoms and cure of a disease on a living body is known as therapeutic effect
- Enzymes: Proteins which perform the role of biological catalysts in the body are called enzymes
- Functions of enzymes:
(i) The first function of an enzyme is to hold the substrate for a chemical reaction. Active sites of enzymes hold the substrate molecule in a suitable position so that it can be attacked by the reagent effectively.
(ii) The second function of an enzyme is to provide functional groups that will attack the substrate and carry out chemical reaction. - Role of drugs: Main role of drugs is to either increase or decrease role of enzyme catalysed reactions. Inhibition of enzymes is a common role of drug action.
- Enzyme inhibitor: Enzyme inhibitor is drug which inhibits catalytic activity of enzymes or blocks the binding site of the enzyme and eventually prevents the binding of substrate with enzyme.
- Drug can inhibit attachment of substrate on active site of enzymes in following ways:
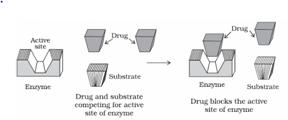
- Competitive Inhibition: Competitive Inhibitors are the drugs that compete with the natural substrate for their attachment on the active sites of enzymes.
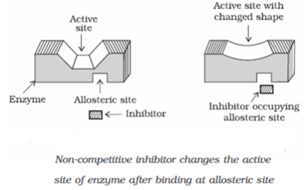
- Non-Competitive Inhibition: Some drugs do not bind to the enzyme’s active site, instead bind to a different site of enzyme called allosteric site. This binding of inhibitor at allosteric site changes the shape of the active site in such a way that substrate cannot recognise it. If the bond formed between an enzyme and an inhibitor is a strong covalent bond and cannot be broken easily, then the enzyme is blocked permanently. The body then degrades the enzyme-inhibitor complex and synthesizes the new enzyme.
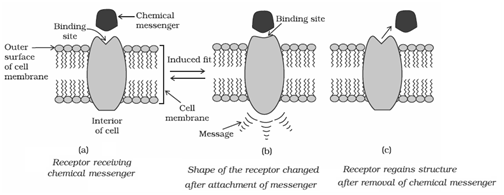
- Receptors: Proteins which are vital for communication system in the body are called receptors. Receptors show selectivity for one chemical messenger over the other because their binding sites have different shape, structure, and amino acid composition.
- Receptors as Drug Targets: In the body, message between two neurons and that between neurons to muscles is communicated through chemical messengers. They are received at the binding sites of receptor proteins. To accommodate a messenger, shape of the receptor site changes which brings about the transfer of message into the cell. Chemical messenger gives message to the cell without entering the cell.
- Antagonists and Agonists: Drugs that bind to the receptor site and inhibit its natural function are called antagonists. These are useful when blocking of message is required. Drugs that mimic the natural messenger by switching on the receptor are called agonists. These are useful when there is lack of natural chemical messenger.
- Therapeutic action of different classes of drugs:
(i) Antacid: Chemical substances which neutralize excess acid in the gastric juices and give relief from acid indigestion, acidity, heart burns and gastric ulcers. Examples: Eno, gelusil, digene etc.
(ii) Antihistamines: Chemical substances which diminish or abolish the effects of histamine released in body and hence prevent allergic reactions. Examples: Brompheniramine (Dimetapp) and terfenadine (Seldane).
(iii) Neurologically Active Drugs: Drugs which have a neurological effect i.e. affects the message transfer mechanism from nerve to receptor. - Tranquilizers: Chemical substances used for the treatment of stress and mild or severe mental diseases. Examples: Derivatives of barbituric acids like veronal, amytal, Nembutal, luminal, seconal.
- Analgesics: Chemical substances used to relieve pain without causing any disturbances in the nervous system like impairment of consciousness, mental confusion, in coordination or paralysis etc.
- Classification of Analgesics:
(a) Non-narcotic analgesics: They are non-addictive drugs. Examples: Aspirin, Ibuprofen, Naproxen, Dichlofenac Sodium.
(b) Narcotic analgesics: When administered in medicinal doses, these drugs relieve pain and produce sleep. Examples: Morphine and its derivatives) Anti-microbials: Drugs that tends to destroy/prevent development or inhibit the pathogenic action of microbes such as bacteria (antibacterial drugs), fungi (anti-fungal agents), virus (antiviral agents), or other parasites (anti-parasitic drugs) selectively.
v) Anti-fertility Drugs: Chemical substances used to prevent conception or fertilization are called anti-fertility drugs. Examples – Norethindrone, ethynylestradiol (novestrol). - Types of antimicrobial drugs :
(a) Antibiotics: Chemical substances produced by microorganisms that kill or prevent the growth of other microbes.
Classification of antimicrobial drugs based on the mode of control of microbial diseases:
- Bactericidal drugs – Drugs that kills organisms in the body. Examples – Penicillin, Aminoglycosides, Ofloxacin.
- Bacteriostatic drugs – Drugs that inhibits growth of organisms. Examples – Erythromycin, Tetracycline, Chloramphenicol.
Classification of antimicrobial drugs based on its spectrum of action:
- Broad-spectrum antibiotics – Antibiotics which kill or inhibit a wide range of Gram-positive and Gram-negative bacteria are called broad-spectrum antibiotics. Examples – Ampicillin and Amoxycillin.
- Narrow spectrum antibiotics – Antibiotics which are effective mainly against Gram-positive or Gram-negative bacteria are called narrow-spectrum antibiotics. Examples- Penicillin G.
- Limited spectrum antibiotics -Antibiotics effective against a single organism or disease
(b) Antiseptics: Chemical substances that kill or prevent growth of microorganisms and can be applied on living tissues such as cuts, wounds etc., are called anti-spetics. Examples – Soframicine, dettoletc.
(c) Disinfectants: Chemical substances that kill microorganisms but cannot be applied on living tissues such as cuts, wounds etc., are called disinfectants. Examples – Chlorine (Cl2), bithional, iodoform etc.
- Food additives: Food additives are the substances added to food to preserve its flavor or improve its taste and appearance.
- Different types of food additives:
- Artificial Sweetening Agents: Chemical compounds which gives sweetening effect to the food and enhance its flavour. Examples – Aspartame, Sucrolose and Alitame.
- Food preservatives: Chemical substances which are added to food material to prevent their spoilage due to microbial growth. Examples – Sugar, Salts, Sodium benzoate
- Food colours: Substances added to food to increase the acceptability and attractiveness of the food product. Examples – Allura Red AC, Tartrazine
- Nutritional supplements: Substances added to food to improve the nutritional value. Examples -Vitamins, minerals etc.
- Fat emulsifiers and stabilizing agents: Substances added to food products to give texture and desired consistency. Examples – Egg yolk (where the main emulsifying chemical is Lecithin)
- Antioxidants: Substances added to food to prevent oxidation of food materials. Examples – ButylatedHydroxy Toluene (BHT), ButylatedHydroxy Anisole (BHA).
- Soaps: It is a sodium or potassium salts of long chain fatty acids like stearic, oleic and palmitic acid.
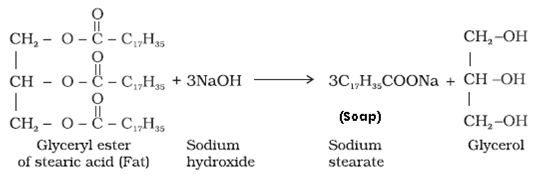
This reaction is known as saponification.
-
Types of soaps:
- Toilet soaps are prepared by using better grades of fats and oil sand care is taken to remove excess alkali. Colour and perfumes are added to make these more attractive.
- Transparent soaps are made by dissolving the soap in ethanol and then evaporating the excess solvent.
- In medicated soaps, substances of medicinal value are added. In some soaps, deodorants are added.
- Shaving soaps contain glycerol to prevent rapid drying. A gum called, rosin is added while making them. It forms sodium rosinate which lathers well.
- Laundry soaps contain fillers like sodium rosinate, sodium silicate, borax, and sodium carbonate.
- Soaps that float in water are made by beating tiny air bubbles before their hardening.
- Soap chips are made by running a thin sheet of melted soap ontoa cool cylinder and scraping off the soaps in small broken pieces.
- Soap granules are dried miniature soap bubbles.
- Soap powders and scouring soaps contain some soap, a scouring agent (abrasive) such as powdered pumice or finely divided sand, and builders like sodium carbonate and trisodium phosphate.
- Advantages of using soaps: Soap is a good cleansing agent and is 100% biodegradable i.e. micro- organisms present in sewage water can completely oxidize soap. Therefore, soaps do not cause any pollution problems.
- Disadvantages of using soaps: Soaps cannot be used in hard water because hard water contains metal ions like Ca2+ and Mg2+ which react with soap to form a white precipitate of calcium and magnesium salts
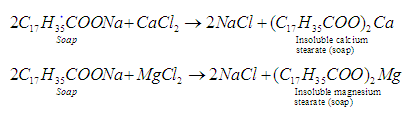
These precipitates stick to the fibers of the cloth as gummy mass and block the ability of soaps to remove oil and grease from fabrics. Therefore, it interferes with the cleansing ability of the soap and makes the cleansing process difficult.
In acidic medium, the acid present in solution precipitate the insoluble free fatty acids which adhere to the fabrics and hence block the ability of soaps to remove oil and grease from the fabrics. Hence soaps cannot be used in acidic medium
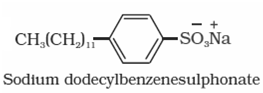
- Detergents: Detergents are sodium salts of long chain of alkyl benzene sulphonic acids or sodium salts of long chain of alkyl hydrogen sulphates.
-
Classification of detergents:
(a)Anionic detergents: Anionic detergents are sodium salts of sulphonated long chain alcohols or hydrocarbons. Alkyl hydrogen sulphates formed by treating long chain alcohols with concentrated sulphuric acid are neutralised with alkali to form anionic detergents. Similarly alkyl benzene sulphonates are obtained by neutralising alkyl benzene sulphonic acids with alkali. Anionic detergents are termed so because a large part of molecule is an anion.
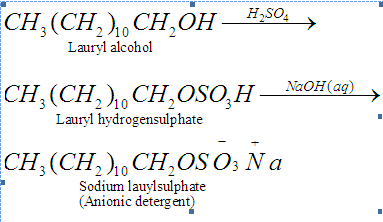
They are used in household cleaning like dishwasher liquids, laundry liquid detergents, laundry powdered detergents etc. They are effective in slightly acidic solutions where soaps do not work efficiently.
(b)Cationic detergents: Cationic detergents are quarternary ammonium salts of mine with acetates, chlorides or bromides as anions. Cationic parts possess a long hydrocarbon chain and a positive charge on nitrogen atom. Cationic detergents are termed so because a large part of molecule is a cation. Since they possess germicidal properties, they are used as germicides. They has strong germicidal action, but are expensive.
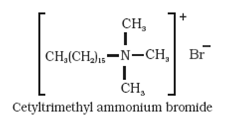
(c) Non- ionic detergents: They do not contain any ion in their constitution. They are like esters of high molecular mass.
Example: Detergent formed by condensation reaction between stearic acid reacts and poly ethyl eneglycol.
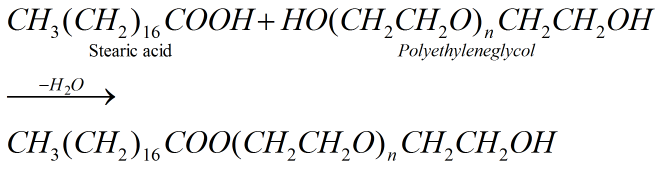
It is used in Making liquid washing detergents. They have effective H- bonding groups at one end of the alkyl chain which make them freely water soluble.
- Biodegradable detergents: Detergents having straight hydrocarbon chains that are easily decomposed by microorganisms. Example: Sodium lauryl sulphate
- Non-Biodegradable detergents: Detergents having branched hydrocarbon chains that are not easily decomposed by microorganisms.
CBSE Class 12 Revision Notes and Short Key Notes
CBSE quick revision note for class 12 Chemistry, Physics Maths, Biology, and other subject are very helpful to revise the whole syllabus during exam days. The revision notes covers all important formulas and concepts given in the chapter. Even if you wish to have an overview of a chapter, quick revision notes are here to do if for you. These notes will certainly save your time during stressful exam days.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Economics
- Business Studies
- Computer Science
- Informatics Practices
- English Core
- History
- Physical Education
To download Chemistry in Everyday Life class 12 Notes Chemistry, sample paper for class 12 Physics, Chemistry, Biology, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies, and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT Chemistry in Everyday Life, NCERT Exemplar Chemistry in Everyday Life, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.
- Solid State class 12 Notes Chemistry
- Solutions class 12 Notes Chemistry
- Electrochemistry class 12 Notes Chemistry
- Chemical Kinetics class 12 Notes Chemistry
- Surface Chemistry class 12 Notes Chemistry
- General Principles and Processes of Isolation of Elements class 12 Notes Chemistry
- The p-Block Elements class 12 Notes Chemistry
- The d- and f- Block Elements class 12 Notes Chemistry
- Coordination Compounds class 12 Notes Chemistry
- Haloalkanes and Haloarenes class 12 Notes Chemistry
- Alcohols Phenols and Ethers class 12 Notes Chemistry
- Aldehydes Ketones and Carboxylic Acids class 12 Notes Chemistry
- Amines class 12 Notes Chemistry
- Biomolecules class 12 Notes Chemistry
- Polymers class 12 Notes Chemistry

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now