CBSE Question Paper 2015 class 12 Chemistry conducted by Central Board of Secondary Education, New Delhi in the month of March 2015. CBSE previous year question papers with the solution are available in the myCBSEguide mobile app and website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
CBSE Question Paper 2015 class 12 Chemistry

Class 12 Chemistry list of chapters
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d and f Block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday life
CBSE Question Paper 2015 class 12 Chemistry
General Instructions:
(i) There ae total of 26 questions in all. All questions are compulsory.
(ii) Question numbers 1 to 5 are very short answers questions carrying 1 mark each.
(iii) Question numbers 6 to 10 are short answers questions carrying 2 marks each.
(iv) Question numbers 11 to 22 are also short answer questions carrying 3 marks each.
(v) Question number 23 is value based question carrying 4 marks.
(vi) Question numbers 24 to 26 are long answer questions carrying 5 marks each.
1. Which stoichiometric defeat in crystals increases the density of a solid?
2. What is meant by ‘shape selective catalysis’ of reactions?
3. Which of the following is more stable complex and why?
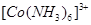 and
and 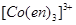
4. Write the IUPAC name of the following compound:
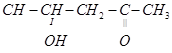
5. Which would undergo 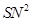 reaction faster in the following pair and why?
reaction faster in the following pair and why?
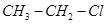 and
and 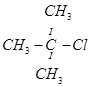
6. State Raoult’s law for the solution containing volatile components. What is the similarity between Raoult’s law and Henry’s law?
OR
Determine the osmotic pressure of a solution prepared by dissolving 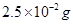 of
of 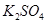 in 2L of water at
in 2L of water at 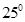 C, assuming that is completely dissociated.
C, assuming that is completely dissociated.
(R = 0.0821 L atm 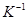
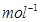 , Molar mass of
, Molar mass of 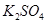 = 174 g mol
= 174 g mol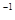 )
)
7. For a first order reaction, show that time required for 99% completion is twice the time required for the completion of 90% of reaction.
8. How do you prepare?
(a) 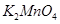 from
from 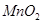 ?
?
(b) 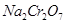 from
from 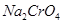 ?
?
9. Write the mechanism of the following reaction:
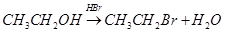
10. Arrange the following in the decreasing order of given property indicated:
(a) 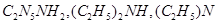 and
and 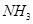
(b) 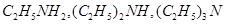 and
and 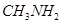
11. (a) What type of semiconductor is obtained when silicon is doped with boron?
(b) What type of magnetism is shown in the following alignment of magnetic moments?

(c) What type of point defect is produced when AgCl is doped with 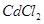 ?
?
OR
Niobium crystallises in body-centred cubic structure. If density is 8.55g 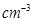 , calculate atomic radius of niobium using its atomic mass as 93u.
, calculate atomic radius of niobium using its atomic mass as 93u.
12. 3.9g of benzoic acid dissolved in 49g of benzene shows a depression in freezing point of 1.62K. Calculate the Van’t Hoff factor and predict the nature of solute (associated or dissociated).
(Given: Molar mass of benzoic acid = 122g 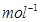 ,
, 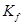 for benzene = 4.9 K kg
for benzene = 4.9 K kg 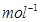 )
)
13. (a) What do you understand by the ‘order of reaction’? Identify the reaction order from each of the following units of reaction rate constant:
(i) 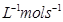
(ii) 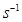
(b) Give one example of zero order reaction.
14. Explain the following terms:
(a) Tyndall effect
(b) Dialysis
(c) Electrophoresis
15. Write the principles of the following methods:
(a) Froth flotation method
(b) Electrolytic refining
(c) Zone refining
16. Complete the balance the following chemical equations:
(a) 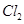 + NaOH (hot and conc.)
+ NaOH (hot and conc.) 
(b) 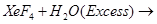
(c) 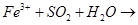
17. (a) Name the element of 3d transition series which shows maximum number of oxidation state.
(b) Name a member of the lanthanoid series which is well-known to exhibit +2 oxidation state.
(c) Complete the equation:
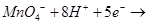
18. (a) Draw the geometrical isomers of complex compound 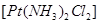
(b) On the basis of crystal field theory, write the electronic configuration for 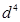 ion if
ion if 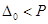
(c) Write the hybridization and magnetic behaviour of the complex compound 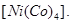
19. Give reasons for the following:
(a) The presence of nitro group 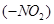 at ortho or Para positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
at ortho or Para positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.
(b) Racemic mixture is optically inactive.
20. Give the structure of A, B and C in the following rections:
(a) 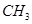 – Br
– Br 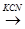 A
A 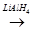 B
B 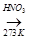 C
C
(b) 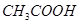
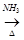 A
A 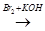 B
B 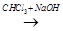 C
C
21. (a) Deficiency of which vitamin causes scurvy?
(b) What type of linkage is responsible for the formation of proteins?
(c) Write the product formed when glucose is treated with HI.
22. (a) What are disinfectants? Give an example.
(b) Give two examples of macromolecules that are chosen as drug targets.
(c) What are anionic detergents? Give an example.
23. After the ban on plastic bags, students of one school decided to make the people aware of the harmful effects of plastic bags on environment and Yamuna river. To make the awareness more impactful, they organized rally by joining hands with other schools and distributed paper bags to vegetable vendors, shopkeepers and departmental stores. All students pledged not to use polythene bags in future to save Yamuna river.
After reading the above passage, answer the following questions:
(a) What values are shown by the students.
(b) What are biodegradable polymers? Give one example.
(c) Is polythene a consideration or an addition polymer.
24. (a) Explain the following observations:
(i) The products of electrolysis of molten NaCl are sodium metal and chlorine gas.
(ii) The products of electrolysis of aqueous sodium chloride are NaOH, 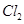 and
and 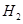
(b) The molar conductivities of 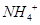 ion and
ion and 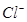 ion are 73.5 s
ion are 73.5 s 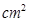
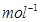 and 76.25 s
and 76.25 s 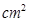
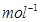 respectively. The specific conductivity of 0.1M
respectively. The specific conductivity of 0.1M 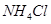 is
is 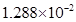 s
s 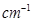 . Calculate the dissociated constant of
. Calculate the dissociated constant of 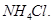
OR
(a) Describe the principle and overall reaction involved in the working of hydrogen-oxygen fuel cell.
(b) Write the Nernst equation and calculate the emf of the following cell at 298K:
Cu(s)/ 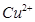 (0.13M)
(0.13M) 
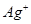 (
(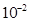 M) /Ag(s)
M) /Ag(s)
25. Assign a possible reason for the following:
(a) Fluorine forms only one oxoacid HOF.
(b) Nitrogen exist as diatomic 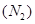 while phosphorus exist as tetratomic
while phosphorus exist as tetratomic 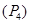 at room temperature.
at room temperature.
(c) The negative value of the electron gain enthalpy of fluorine is less than that of chlorine.
(d) Interhalogen compounds are more reactive than halogens.
(e) 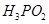 is a monoprotic acid.
is a monoprotic acid.
OR
(a) Give reasons for the following:
(i) 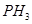 has lower boiling point than
has lower boiling point than 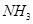
(ii) Oxygen shows catenation behaviour less than Sulphur.
(iii) Helium is used in diving apparatus.
(b) Draw the structures of the following molecules:
(i) 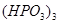
(ii) 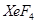
26. (a) Give simple chemical tests to distinguish between the following pairs of compounds:
(i) Proponal and propanone
(ii) Benzaldehyde and acetaldehyde
(b) Write the products of the following reactions:
(i) 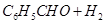 NCONHN
NCONHN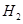
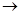
(ii) 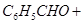 conc. NaOH
conc. NaOH 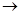
(iii) 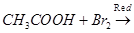
OR
(a) Account for the following:
(i) 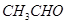 is more reactive than
is more reactive than 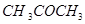 towards reaction with HCN.
towards reaction with HCN.
(ii) Carboxylic acid is stronger acid than phenol.
(b) Identify A, B and C in the following sequence of reactions:
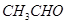
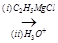 A
A 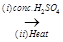 B
B 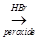 C
C
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
Chemistry Question Paper 2015
Download class 12 Chemistry question paper from best CBSE App the myCBSEguide. CBSE class 12 Chemistry question paper 2015 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper. CBSE question papers 2015 for class 12 Chemistry have 26 questions with solution.
Previous Year Question Paper for class 12 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 209, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Business Studies
- Economics
- History
- Geography
- Political Science
- Physical Education
- Computer Science
- Informatics Practices
- English Core
- Hindi Core
- Hindi Elective
- Other Subjects
To download CBSE Question Paper 2015 class 12 Accountancy, Chemistry, Physics, History, Political Science, Economics, Geography, Computer Science, Home Science, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now