CBSE Question Paper 2014 class 12 Chemistry conducted by Central Board of Secondary Education, New Delhi in the month of March 2014. CBSE previous year question papers with the solution are available in the myCBSEguide mobile app and website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
CBSE Question Paper 2014 class 12 Chemistry

Class 12 Chemistry list of chapters
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d and f Block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday life
CBSE Question Paper 2014 class 12 Chemistry
General Instructions
- All questions are compulsory.
- Marks for each question are indicated against it.
- Question numbers 1 to 8 are very short-answer questions, carrying 1 mark each. Answer these in one word or about one sentence each.
- Question numbers 9 to 18 are short-answer questions, carrying 2 marks each. Answer these in about 30 words each.
- Question numbers 19 to 27 are short-answer questions of 3 marks each. Answer these in about 40 words each.
- Question numbers 28 to 30 are long-answer questions of 5 marks each. Answer these in about 70 words each.
- Use Log Tables, if necessary Use of calculators is not permitted.
1. Name the depressant which is used to separate ZnS and PbS ores in froth floatation process. [1]
2. Based on the type of dispersed phase, what type of colloid is micelles? [1]
3. Arrange the following in the increasing order of their basic character: [1]
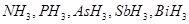
4. What type of isomerism is exhibited by the following complex: [1]
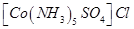
5. Write IUPAC name of the following compound:[1]
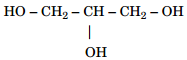
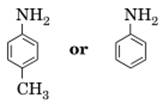
6. Which of the two is more basic and why? [1]
7. What are biocatalysts? Give an example. [1]
8. Express the relationship between atomic radius (r) and the edge length (a) in the b.c.c. unit cell. [1]
9. Write the role of the following: [2]
(i) Iodine in the refining of zirconium
(ii) Silica in the extraction of copper from copper matte
10. Name the two most important allotropes of sulphur. Which one of the two is stable at room temperature? What happen when the stable form is heated above 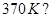 [2]
[2]
Or
(i) Write the conditions to maximize the yield of 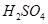 by contact process.
by contact process.
(ii) Why is 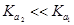 for
for 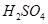 in water? [2]
in water? [2]
11. Complete the following equations: [2]
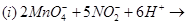
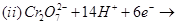
12. Write the state of hybridization, shape and IUPAC name of the complex 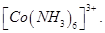
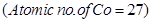 [2]
[2]
13. Write chemical equations when
(i) ethyl chloride is treated with alcoholic 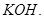
(ii) chlorobenzene is treated with 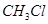 in the presence of anhydrous
in the presence of anhydrous 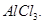 [2]
[2]
14. Write the mechanism of the following reaction: [2]
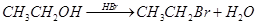
15. Name the different reagents needed to perform the following reactions: [2]
(i) Phenol to Benzene
(ii) Dehydration of propan-2-ol to propene
(iii) Friedel − Crafts acetylation of anisole
(iv) Dehydrogenation of ethanol to ethanol
16. A solution of 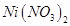 is electrolyzed between platinum electrodes using a current of
is electrolyzed between platinum electrodes using a current of 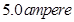 for
for 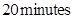 . What mass of nickel will be deposited at the cathode? [2]
. What mass of nickel will be deposited at the cathode? [2]
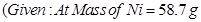
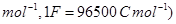
17. Define half-life of a reaction. Write the expression of half-life for [2]
(i) zero order reaction and
(ii) first order reaction.
18. (a) Which alkyl halide from the following pairs would you expect to react more rapidly by an 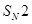 mechanism and why?
mechanism and why?
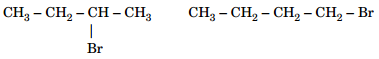
(b) Racemisation occurs in 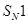 reactions. why? [2]
reactions. why? [2]
19. The following data were obtained during the first order thermal decomposition of 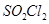 at a constant volume: [3]
at a constant volume: [3]
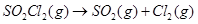
| Experiment | 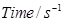 |
Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
20. (i) What type of stoichiometric defect is shown by 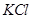 and why?
and why?
(ii) What type of semiconductor is formed when silicon is doped with As?
(iii) Which one of the following is an example of molecular solid:
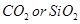
(iv) What type of substances would make better magnets, ferromagnetic or ferrimagnetic? [3]
21. (i) Write two advantages of 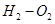 fuel cell over ordinary cell.
fuel cell over ordinary cell.
(ii) Equilibrium constant 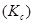 for the given cell reaction is 10. Calculate
for the given cell reaction is 10. Calculate 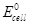 .
.
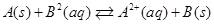 [3]
[3]
22. After the ban on plastic bags, students of one school decided to create awareness among the people about the harmful effects of plastic bags on the environment and the Yamuna river. To make it more impactful, they organized a rally by joining hands with other schools and distributed paper bags to vegetable vendors, shopkeepers and departmental stores. All students pledged not to use polythene bags in future to save the Yamuna river. [3]
After reading the above passage, answer the following questions:
(i) What values are shown by the students?
(ii) What are biodegradable polymers? Give one example.
(iii) Is polythene a homopolymer or copolymer?
23. (i) Define Antihistamine with an example.
(ii) Which one of the following drugs is an antibiotic:
Morphine, Equanil, Chloramphenicol, Aspirin.
(iii) Why is use of aspartame limited to cold food and drink? [3]
24. Define the following terms: [3]
(a) Invert sugar
(b) Vitamins
(c) Nucleoside
25. (a) What are the different oxidation states exhibited by the lanthanoids?
(b) Write two characteristics of the transition elements.
(c) Which of the 3d-block elements may not be regarded as the transition elements and why? [3]
OR
Assign suitable reasons for the following:
(a) The 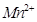 compounds are more stable than
compounds are more stable than 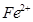 towards oxidation to their +3 state.
towards oxidation to their +3 state.
(b) In the 3d series from 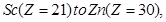 the enthalpy of atomization of
the enthalpy of atomization of 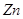 is the lowest.
is the lowest.
(c) 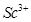 is colourless in aqueous solution whereas
is colourless in aqueous solution whereas 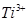 is coloured. [3]
is coloured. [3]
26. (a) What are the different oxidation states exhibited by the lanthanoids?
(b) Write two characteristics of the transition elements.
(c) Which of the 3d-block elements may not be regarded as the transition elements and why? [3]
OR
Assign suitable reasons for the following:
(a) The 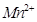 compounds are more stable than
compounds are more stable than 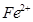 towards oxidation to their +3 state.
towards oxidation to their +3 state.
(b) In the 3d series from 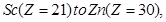 the enthalpy of atomization of
the enthalpy of atomization of 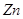 is the lowest.
is the lowest.
(c) 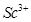 is colourless in aqueous solution whereas
is colourless in aqueous solution whereas 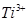 is coloured. [3]
is coloured. [3]
26. (a) Write the expression for the Freundlich adsorption isotherm for the adsorption of gases on solids, in the form of an equation.
(b) What are the dispersed phase and dispersion medium of butter?
(c) A delta is formed at the meeting place of sea and river water. Why? [3]
27. Give the structures of A, B and C in the following reactions: [3]
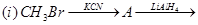
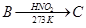
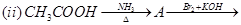
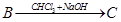
28. (a) Account for the following:
(i) 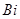 is strong oxidizing agent in the + 5 state.
is strong oxidizing agent in the + 5 state.
(ii) 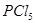 is known but
is known but 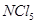 is not known.
is not known.
(iii) Iron dissolves in 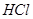 to form
to form 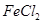 and not
and not 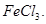
(b) Draw the structures of the following:
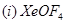
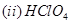 [5]
[5]
Or
(a) Draw the structures of the following:
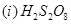
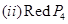
(b) Account for the following:
(i) Sulphur in vapour state exhibits paramagnetism.
(ii) Unlike xenon, no distinct chemical compound of helium is known.
(iii) 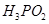 is a stronger reducing agent than
is a stronger reducing agent than 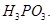 [5]
[5]
29. (a) Write the products formed when ethanal reacts with the following reagents:
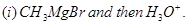
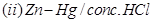
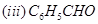 in the presence of dilute
in the presence of dilute 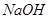 .
.
(b) Give simple chemical tests to distinguish between the following pairs of compounds:
(i) Benzoic acid and Ethyl benzoate
(ii) Propanal and Butan-2-one [5]
OR
(a) Account for the following:
(i) 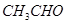 is more reactive than
is more reactive than 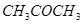 towards reaction with
towards reaction with 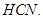
(ii) There are two 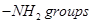 in semicarbazide
in semicarbazide 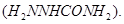 However, only on eis involved in the formation of semicarbazone.
However, only on eis involved in the formation of semicarbazone.
(b) Write the chemical equation to illustrate each of the following name reactions:
(i) Rosenmund reduction
(ii) Hell-Volhard-Zelinsky reaction
(iii) Cannizzaro reaction [5]
30. (a) State Raoult’s law for a solution containing volatile components. Name the solution which follows Raoult’s law at all concentrations and temperatures. [2]
(b) Calculate the boiling point elevation for a solution prepared by adding 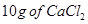 to
to 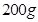 of water.
of water. 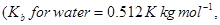
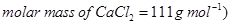 [3]
[3]
Or
(a) Define the following terms: [3]
(i) Azeotrope
(ii) Osmotic pressure
(iii) Colligative properties
(b) Calculate the molarity of 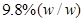 solution of
solution of 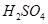 if the density of the solution is
if the density of the solution is 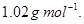
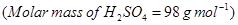 [2]
[2]
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
Chemistry Question Paper 2014
Download class 12 Chemistry question paper with solution from best CBSE App the myCBSEguide. CBSE class 12 Chemistry question paper 2014 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper. CBSE question papers 2014 for class 12 Chemistry have 30 questions with solution.
Previous Year Question Paper for class 12 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 209, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Business Studies
- Economics
- History
- Geography
- Political Science
- Physical Education
- Computer Science
- Informatics Practices
- English Core
- Hindi Core
- Hindi Elective
- Other Subjects
To download CBSE Question Paper 2014 class 12 Accountancy, Chemistry, Physics, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now