CBSE Question Paper 2013 class 12 Chemistry conducted by Central Board of Secondary Education, New Delhi in the month of March 2013. CBSE previous year question papers with the solution are available in the myCBSEguide mobile app and website. The Best CBSE App for students and teachers is myCBSEguide which provides complete study material and practice papers to CBSE schools in India and abroad.
CBSE Question Paper 2013 class 12 Chemistry

Class 12 Chemistry list of chapters
- The Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- General Principles and Processes of Isolation of Elements
- The p-Block Elements
- The d and f Block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Amines
- Biomolecules
- Polymers
- Chemistry in Everyday life
CBSE Question Paper 2013 class 12 Chemistry
General Instructions
- All questions are compulsory.
- Marks for each question are indicated against it.
- Question numbers 1 to 8 are very short-answer questions, carrying 1 mark each. Answer these in one word or about one sentence each.
- Question numbers 9 to 18 are short-answer questions, carrying 2 marks each. Answer these in about 30 words each.
- Question numbers 19 to 27 are short-answer questions of 3 marks each. Answer these in about 40 words each.
- Question numbers 28 to 30 are long-answer questions of 5 marks each. Answer these in about 70 words each.
- Use Log Tables, if necessary Use of calculators is not permitted.
CHEMISTRY 2013 (Delhi)
1. How many atoms constitute one unit cell of a face-centered cubic crystal? [1]
2. Name the method used for the refining of Nickel metal. [1]
3. What is the covalency of nitrogen in 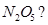 [1]
[1]
4. Write the IUPAC name of [1]
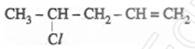
5. What happens when 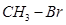 is treated with
is treated with 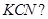 [1]
[1]
6. Write the structure of 3-methyl butanal. [1]
7. Arrange the following in increasing order of their basic strength in aqueous solution: [1]
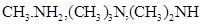
8. What are three types of RNA molecules which perform different functions? [1]
9. 18g of glucose, 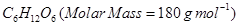 is dissolved in 1Kg of water in a sauce pan. At what temperature will this solution boil? [2]
is dissolved in 1Kg of water in a sauce pan. At what temperature will this solution boil? [2]
10. The conductivity of 0.20 M solution of 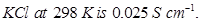 Calculate its molar conductivity. [2]
Calculate its molar conductivity. [2]
11. Write the dispersed phase and dispersion medium of the following colloidal system: [2]
(i) Smoke
(ii) Milk
OR
What are lyophilic and lyophobic colloids? Which of these sols can be easily coagulated on the addition of small amounts of electrolytes? [2]
12. Write the differences between physisorption and chemisorption with respect to the following: [2]
(i) Specificity
(ii) Temperature dependence
(iii) Reversibility and
(iv) Enthalpy change
13. (a) Which solution is used for the leaching of silver metal in the presence of air in the metallurgy of silver?
(b) Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore? [2]
14. What happens when
(i) 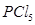 is heated?
is heated?
(ii) 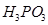 is heated?
is heated?
Write the reaction involved.
15. (a) Which metal in the first transition series (3d series) exhibits +1 oxidation state most frequently and why?
(b) Which of the following cations are colored in aqueous solutions and why?
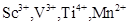
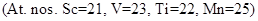 [2]
[2]
16. Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same. [2]
17. Explain the mechanism of the following reaction: [2]
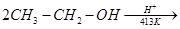
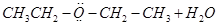
18. How will you convert: [2]
(i) Propene to Propan-2-ol?
(ii) Phenol to 2, 4, 6 – trinitrophenol?
19. (a) What type of semiconductor is obtained when silicon is doped with boron?
(b) What type of magnetism is shown in the following alignment of magnetic moments?

(c) What type of point defect is produced when 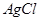 is doped with
is doped with 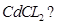 [3]
[3]
20. Determine the osmotic pressure of a solution prepared by dissolving 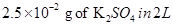 of water
of water 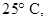 assuming that it is completely dissociated.
assuming that it is completely dissociated.
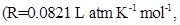
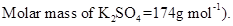 [3]
[3]
21. Calculate the emf of the following cell at 298 K: [3]
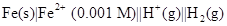
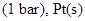
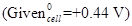
22. How would you account for the following? [3]
(i) Transition metals exhibit variable oxidation states.
(ii) 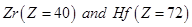 have almost identical radii.
have almost identical radii.
(iii) Transition metals and their compounds act as catalyst.
OR
Complete the following chemical equations:
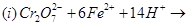
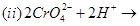
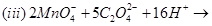
23. Write the IUPAC names of the following coordination compounds: [3]
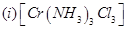
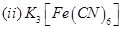
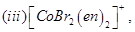
24. Give the structures of A, B and C in the following reactions: [3]
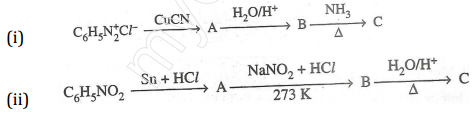
25. Write the names and structures of the monomers of the following polymers: [3]
(i) Buna-S
(ii) Neoprene
(iii) Nylone-6, 6
26. After watching a programme on TV about the adverse effects of junk food and soft drinks on the health of school children, Sonali, a student of Class XII, discussed the issue with the school principal. Principal immediately instructed the canteen contractor to replace the fast food with the fibre and vitamins rich food like sprouts, salad, fruits etc. This decision was welcomed by the parents and the students.
After reading the above passage, answer the following questions:
(a) What values are expressed by Sonali and the Principal of the school?
(b) Give two examples of water-soluble vitamins. [3]
27. (a) Which one of the following is a food preservative?
Equanil, Morphine, Sodium benzoate
(b) Why is bithional added to soap?
(c) Which class of drugs is used in sleeping pills? [3]
28. (a) A reaction is second order in A and first order in B.
(i) Write the differential rate equation.
(ii) How is the rate affected on increasing the concentration of A three times?
(iii) How is the rate affected when the concentration of both A and B are doubled?
(b) A first order reaction takes 40 minutes for 30% decomposition. Calculate 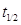 for this reaction.
for this reaction.
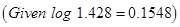
OR
(a) For a first order reaction, show that time required for  completion is twice the time required for the completion of
completion is twice the time required for the completion of  of reaction.
of reaction.
(b) Rate constant ‘k’ of a reaction varies with temperature ‘T’ according to the equation:
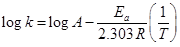
Where 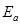 is the activation energy. When a graph is plotted for log k Vs.
is the activation energy. When a graph is plotted for log k Vs. 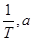 straight line with a slope of
straight line with a slope of 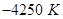 is obtained. Calculated
is obtained. Calculated 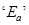 for the reaction.
for the reaction. 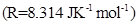 [5]
[5]
29. (a) Give reasons for the following:
(i) Bond enthalpy of 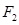 is lower than that of
is lower than that of 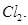
(ii) 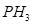 has lower boiling point than
has lower boiling point than 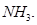
(b) Draw the structures of the following molecules:
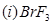
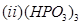
OR
(a) Account for the following:
(i) Helium is used in diving apparatus.
(ii) Fluorine does not exhibit positive oxidation state.
(iii) Oxygen shows catenation behavior less than sulphur.
(b) Draw the structures of the following molecules.

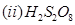 [5]
[5]
30. (a) Although phenoxide ion has more number of resonating structures than Carboxylate ion, Carboxylic acid is a stronger acid than phenol. Give two reasons.
(b) How will you bring about the following conversions?
(i) Propanone to propane
(ii) Benzoyl chloride to benzaldehyde
(iii) Ethanal to but-2-enal
OR
(a) Complete the following reactions:
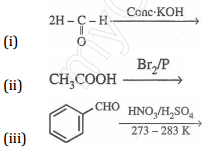
(b) Give simple chemical tests to distinguish between the following pairs of compounds:
(i) Ethanal and Propanal
(ii) Benzoic acid and Phenol [5]
These are questions only. To view and download complete question paper with solution install myCBSEguide App from google play store or login to our student dashboard.
Chemistry Question Paper 2013
Download class 12 Chemistry question paper with solution from best CBSE App the myCBSEguide. CBSE class 12 Chemistry question paper 2013 in PDF format with solution will help you to understand the latest question paper pattern and marking scheme of the CBSE board examination. You will get to know the difficulty level of the question paper. CBSE question papers 2013 for class 12 Chemistry have 30 questions with solution.
Previous Year Question Paper for class 12 in PDF
CBSE question papers 2018, 2017, 2016, 2015, 2014, 2013, 2012, 2011, 2010, 209, 2008, 2007, 2006, 2005 and so on for all the subjects are available under this download link. Practicing real question paper certainly helps students to get confidence and improve performance in weak areas.
- Physics
- Chemistry
- Mathematics
- Biology
- Accountancy
- Business Studies
- Economics
- History
- Geography
- Political Science
- Physical Education
- Computer Science
- Informatics Practices
- English Core
- Hindi Core
- Hindi Elective
- Other Subjects
To download CBSE Question Paper 2013 class 12 Accountancy, Chemistry, Physics, History, Political Science, Economics, Geography, Computer Science, Home Science, Accountancy, Business Studies and Home Science; do check myCBSEguide app or website. myCBSEguide provides sample papers with solution, test papers for chapter-wise practice, NCERT solutions, NCERT Exemplar solutions, quick revision notes for ready reference, CBSE guess papers and CBSE important question papers. Sample Paper all are made available through the best app for CBSE students and myCBSEguide website.

Test Generator
Create question paper PDF and online tests with your own name & logo in minutes.
Create Now
Learn8 App
Practice unlimited questions for Entrance tests & government job exams at ₹99 only
Install Now